QUESTION IMAGE
Question
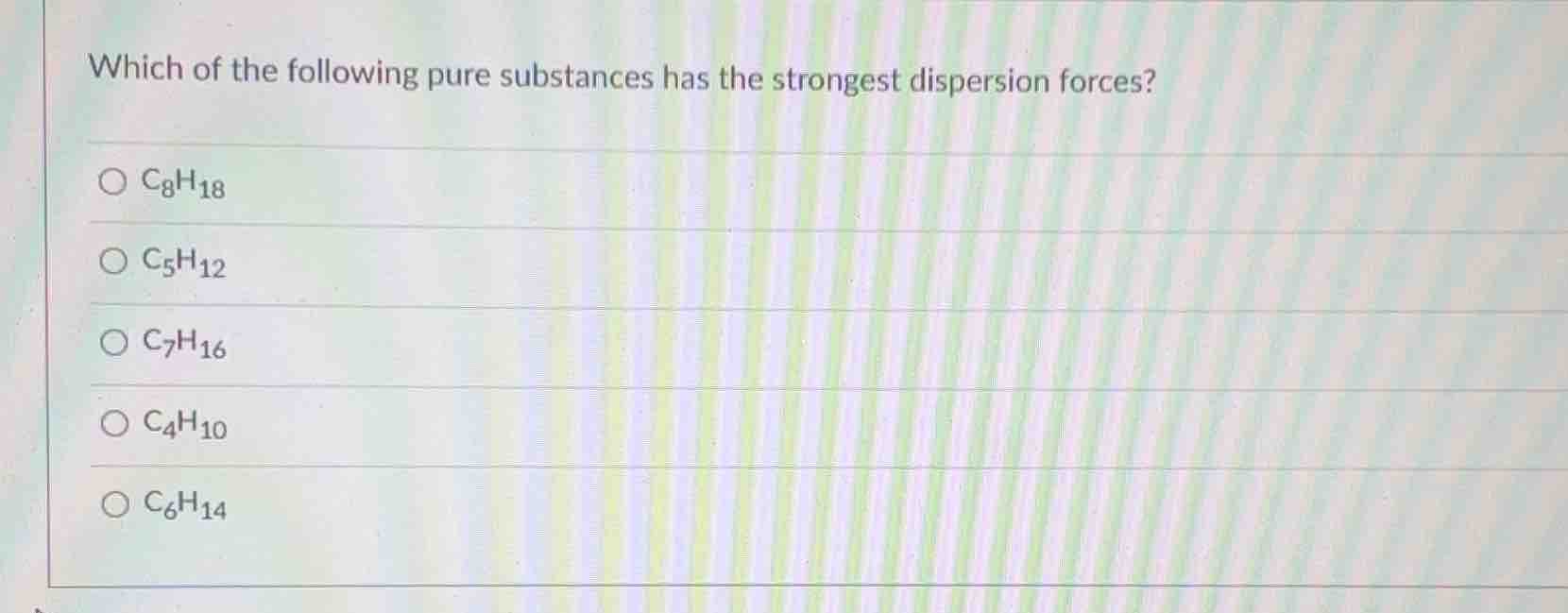
which of the following pure substances has the strongest dispersion forces?
\\(\ce{c8h18}\\)
\\(\ce{c5h12}\\)
\\(\ce{c7h16}\\)
\\(\ce{c4h10}\\)
\\(\ce{c6h14}\\)
Dispersion forces (London dispersion forces) are a type of intermolecular force that depends on the molar mass (or molecular size) of the substance. For non - polar molecules (like the alkanes \(C_nH_{2n + 2}\) here), as the molar mass increases, the number of electrons increases, and the dispersion forces become stronger.
We calculate the molar mass of each alkane:
- For \(C_4H_{10}\): Molar mass \(M=4\times12.01 + 10\times1.008=48.04+10.08 = 58.12\space g/mol\)
- For \(C_5H_{12}\): \(M = 5\times12.01+12\times1.008=60.05 + 12.096=72.146\space g/mol\)
- For \(C_6H_{14}\): \(M=6\times12.01+14\times1.008 = 72.06+14.112 = 86.172\space g/mol\)
- For \(C_7H_{16}\): \(M=7\times12.01+16\times1.008=84.07 + 16.128=100.198\space g/mol\)
- For \(C_8H_{18}\): \(M=8\times12.01+18\times1.008=96.08+18.144 = 114.224\space g/mol\)
Among \(C_4H_{10}\), \(C_5H_{12}\), \(C_6H_{14}\), \(C_7H_{16}\), and \(C_8H_{18}\), \(C_8H_{18}\) has the highest molar mass. So it will have the strongest dispersion forces.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(C_8H_{18}\)