QUESTION IMAGE
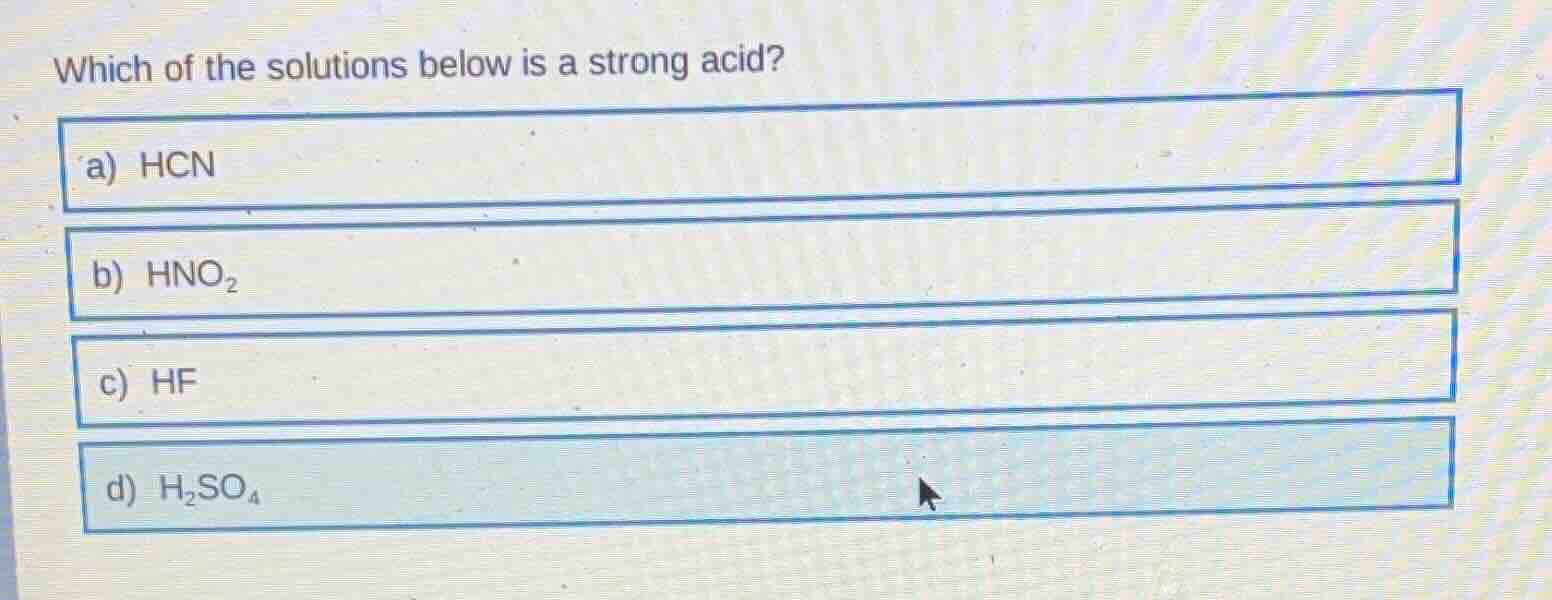
Question
which of the solutions below is a strong acid?
a) hcn
b) hno₂
c) hf
d) h₂so₄
Brief Explanations
Strong acids completely dissociate in aqueous solution. HCN, HNO₂, and HF are weak acids that only partially ionize. H₂SO₄ (sulfuric acid) is a well-known strong acid that fully dissociates in its first proton donation, and its second proton also dissociates to a large extent in dilute solutions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d) $\ce{H2SO4}$