QUESTION IMAGE
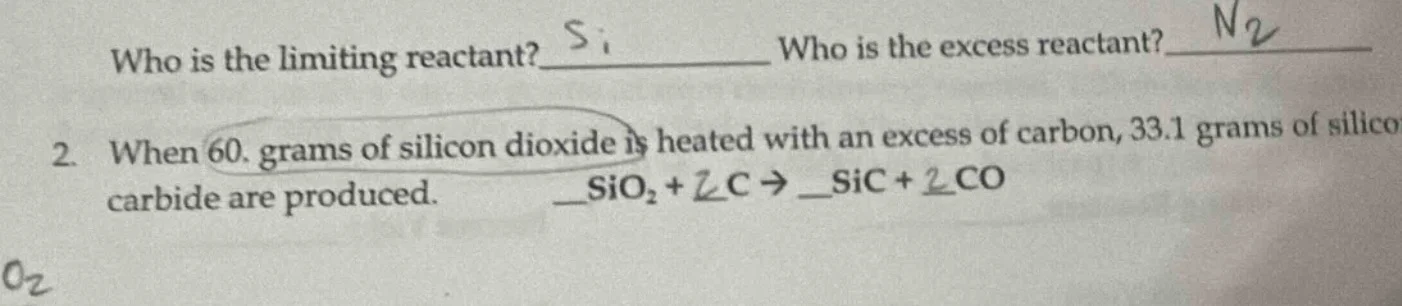
Question
who is the limiting reactant? ____ who is the excess reactant? ____
- when 60. grams of silicon dioxide is heated with an excess of carbon, 33.1 grams of silicon carbide are produced.
sio₂ + c → sic + co
Step1: Balancear la ecuación química
La ecuación balanceada es:
Step2: Calcular masa molar de SiO₂
Masa molar de $\text{SiO}_2$: $M(\text{Si}) + 2 \times M(\text{O}) = 28.09\ \text{g/mol} + 2 \times 16.00\ \text{g/mol} = 60.09\ \text{g/mol}$
Step3: Calcular moles de SiO₂ iniciales
$$n(\text{SiO}_2) = \frac{m}{M} = \frac{60.\ \text{g}}{60.09\ \text{g/mol}} \approx 0.9985\ \text{mol}$$
Step4: Relacionar moles de SiO₂ y SiC
Por estequiometría, 1 mol de $\text{SiO}_2$ produce 1 mol de $\text{SiC}$, así que moles teóricos de $\text{SiC}$: $n(\text{SiC})_{\text{teórico}} = 0.9985\ \text{mol}$
Step5: Calcular masa teórica de SiC
Masa molar de $\text{SiC}$: $28.09\ \text{g/mol} + 12.01\ \text{g/mol} = 40.10\ \text{g/mol}$
$$m(\text{SiC})_{\text{teórico}} = n \times M = 0.9985\ \text{mol} \times 40.10\ \text{g/mol} \approx 40.04\ \text{g}$$
Step6: Calcular rendimiento porcentual (si se requiere)
$$\text{Rendimiento} = \frac{m(\text{SiC})_{\text{real}}}{m(\text{SiC})_{\text{teórico}}} \times 100\% = \frac{33.1\ \text{g}}{40.04\ \text{g}} \times 100\% \approx 82.7\%$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Ecuación química balanceada: $\boldsymbol{\text{SiO}_2 + 3\text{C}
ightarrow \text{SiC} + 2\text{CO}}$
- Reactivo limitante: $\boldsymbol{\text{SiO}_2}$ (el carbono está en exceso)
- Masa teórica de silicuro de carbono: $\boldsymbol{\approx 40.0\ \text{g}}$
- Rendimiento porcentual de la reacción: $\boldsymbol{\approx 82.7\%}$