QUESTION IMAGE
Question
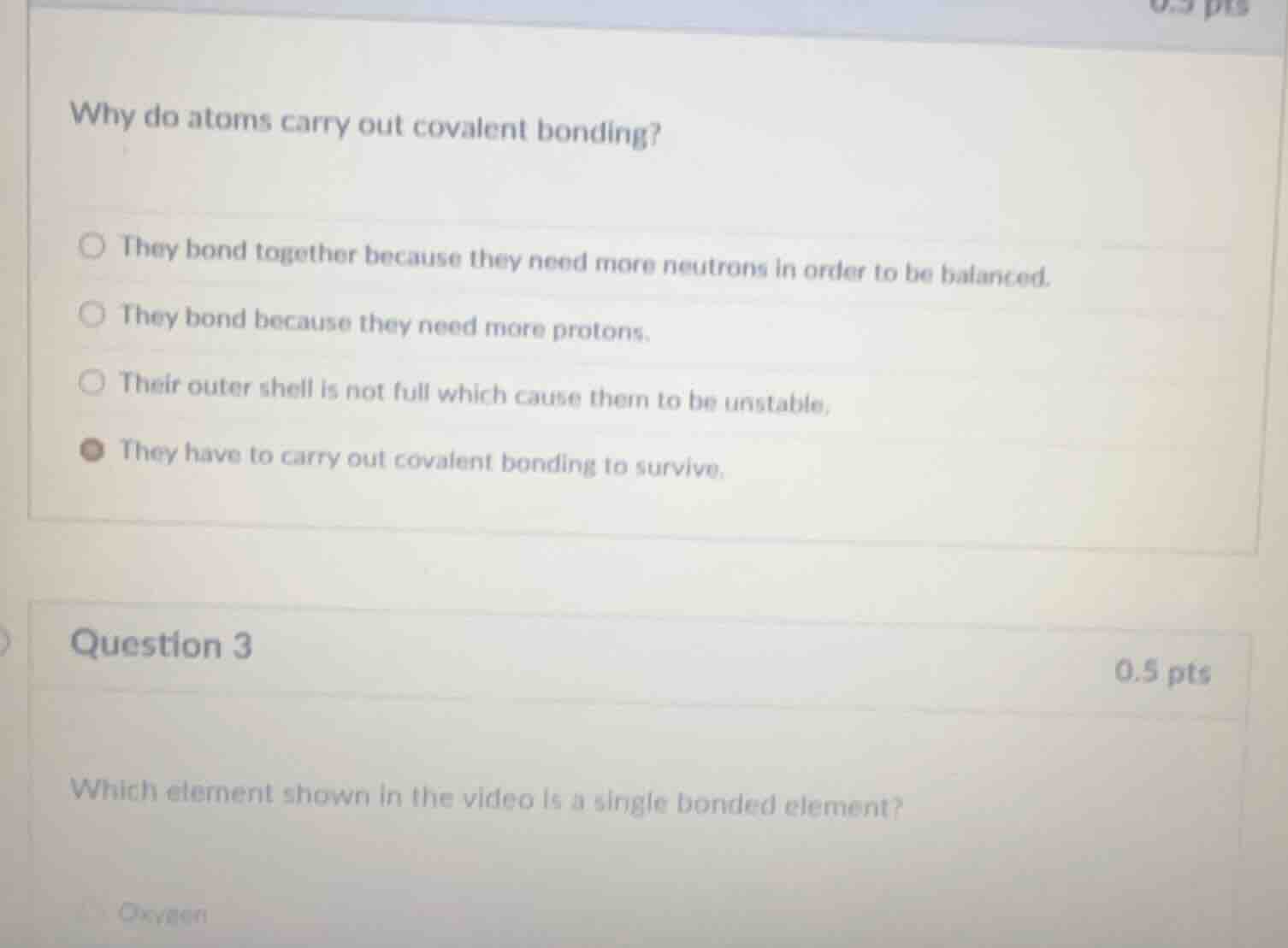
why do atoms carry out covalent bonding?
they bond together because they need more neutrons in order to be balanced.
they bond because they need more protons.
their outer shell is not full which cause them to be unstable.
they have to carry out covalent bonding to survive.
question 3
0.5 pts
which element shown in the video is a single bonded element?
oxygen
Response
Question 1 (Why do atoms carry out covalent bonding?)
Brief Explanations
- Option 1: Covalent bonding is not about neutrons (neutrons are in the nucleus, bonding involves electrons), so incorrect.
- Option 2: Protons are in the nucleus and determine the element, not covalent bonding (which involves electrons), so incorrect.
- Option 3: Atoms form covalent bonds because their outermost electron shell is incomplete (unstable), so they share electrons to fill it, this is correct.
- Option 4: Atoms don't "need" to bond to "survive" (a non - scientific description), so incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The correct option is: Their outer shell is not full which cause them to be unstable.