QUESTION IMAGE
Question
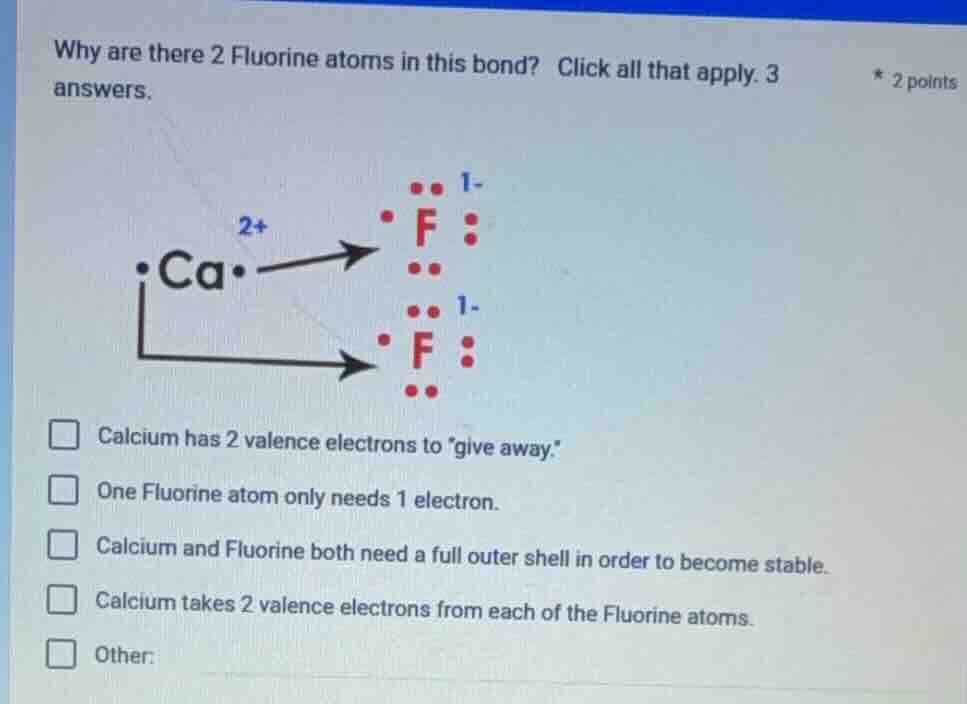
why are there 2 fluorine atoms in this bond? click all that apply. 3 answers.
- 2 points
calcium has 2 valence electrons to give away.
one fluorine atom only needs 1 electron.
calcium and fluorine both need a full outer shell in order to become stable.
calcium takes 2 valence electrons from each of the fluorine atoms.
other:
Brief Explanations
- Calcium is a group 2 metal, so it has 2 valence electrons that it loses to achieve a stable full outer shell.
- Fluorine is a group 17 nonmetal, so it only needs 1 additional electron to complete its outer shell.
- All atoms form bonds to reach a stable full outer electron shell (octet rule for main group elements).
- Calcium loses electrons, it does not take them from fluorine, so that option is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Calcium has 2 valence electrons to "give away."
- One Fluorine atom only needs 1 electron.
- Calcium and Fluorine both need a full outer shell in order to become stable.