QUESTION IMAGE
Question
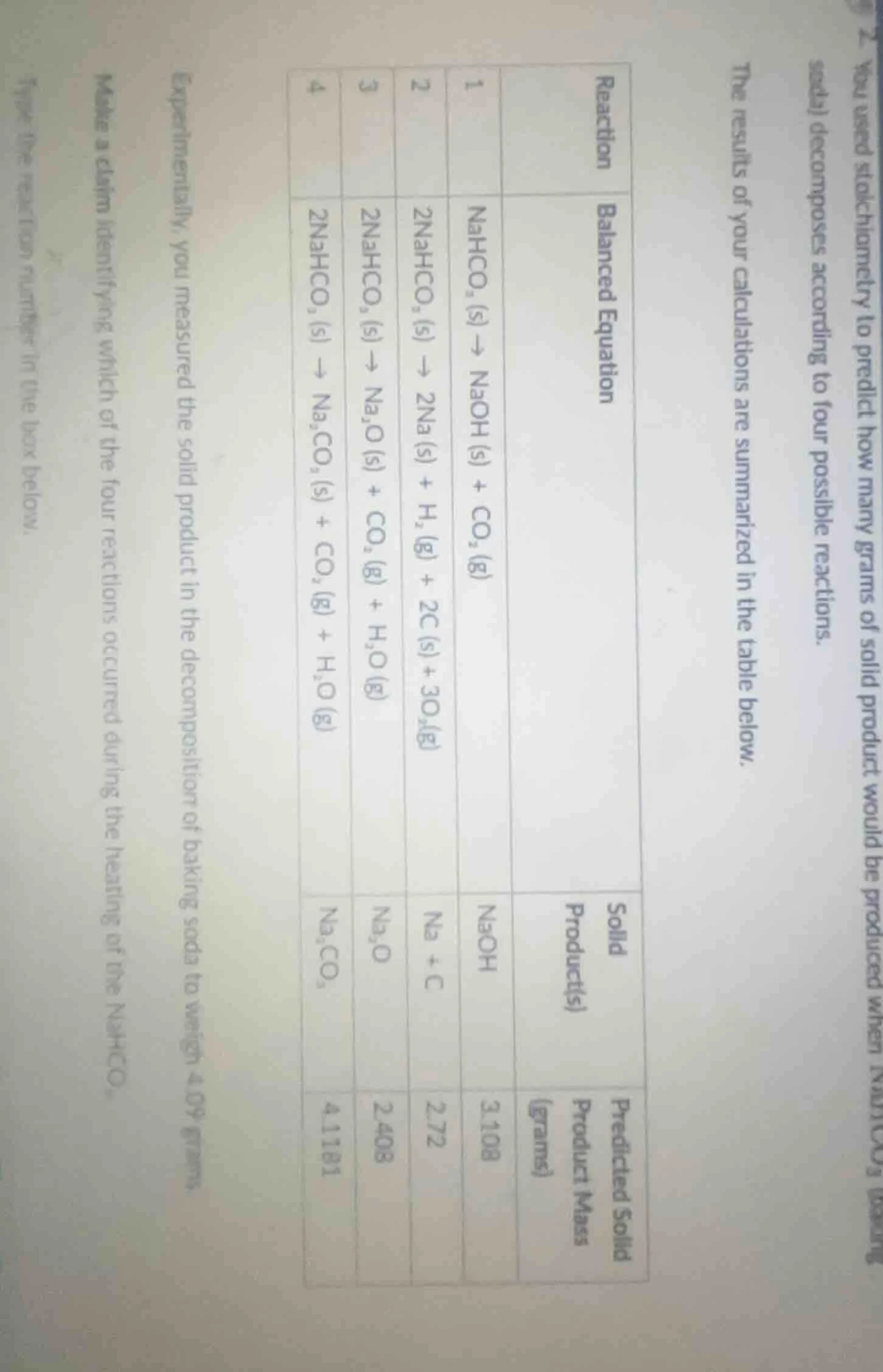
- you used stoichiometry to predict how many grams of solid product would be produced when nahco₃ (baking soda) decomposes according to four possible reactions.the results of your calculations are summarized in the table below.| reaction | balanced equation | solid product(s) | predicted solid product mass (grams) || ---- | ---- | ---- | ---- || 1 | $\text{nahco}_3\text{ (s)} \
ightarrow \text{naoh (s)} + \text{co}_2\text{ (g)}$ | naoh | 3.108 || 2 | $2\text{nahco}_3\text{ (s)} \
ightarrow 2\text{na (s)} + \text{h}_2\text{ (g)} + 2\text{c (s)} + 3\text{o}_2\text{(g)}$ | na + c | 2.72 || 3 | $2\text{nahco}_3\text{ (s)} \
ightarrow \text{na}_2\text{o (s)} + \text{co}_2\text{ (g)} + \text{h}_2\text{o (g)}$ | $\text{na}_2\text{o}$ | 2.408 || 4 | $2\text{nahco}_3\text{ (s)} \
ightarrow \text{na}_2\text{co}_3\text{ (s)} + \text{co}_2\text{ (g)} + \text{h}_2\text{o (g)}$ | $\text{na}_2\text{co}_3$ | 4.1181 |experimentally, you measured the solid product in the decomposition of baking soda to weigh 4.09 grams.make a claim identifying which of the four reactions occurred during the heating of the nahco₃.type the reaction number in the box below.
Step1: Compare experimental vs predicted mass
Experimental mass = 4.09 g; compare to predicted values:
Reaction1: 3.108 g, Reaction2: 2.72 g, Reaction3: 2.408 g, Reaction4: 4.1181 g
Step2: Identify closest predicted value
Calculate difference for Reaction4: $|4.09 - 4.1181| = 0.0281$ g, which is the smallest difference.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Reaction 4 (the decomposition of $\text{NaHCO}_3$ to $\text{Na}_2\text{CO}_3$, $\text{CO}_2$, and $\text{H}_2\text{O}$) occurred, as its predicted solid product mass (4.1181 g) is the closest to the experimentally measured 4.09 g.