QUESTION IMAGE
Question
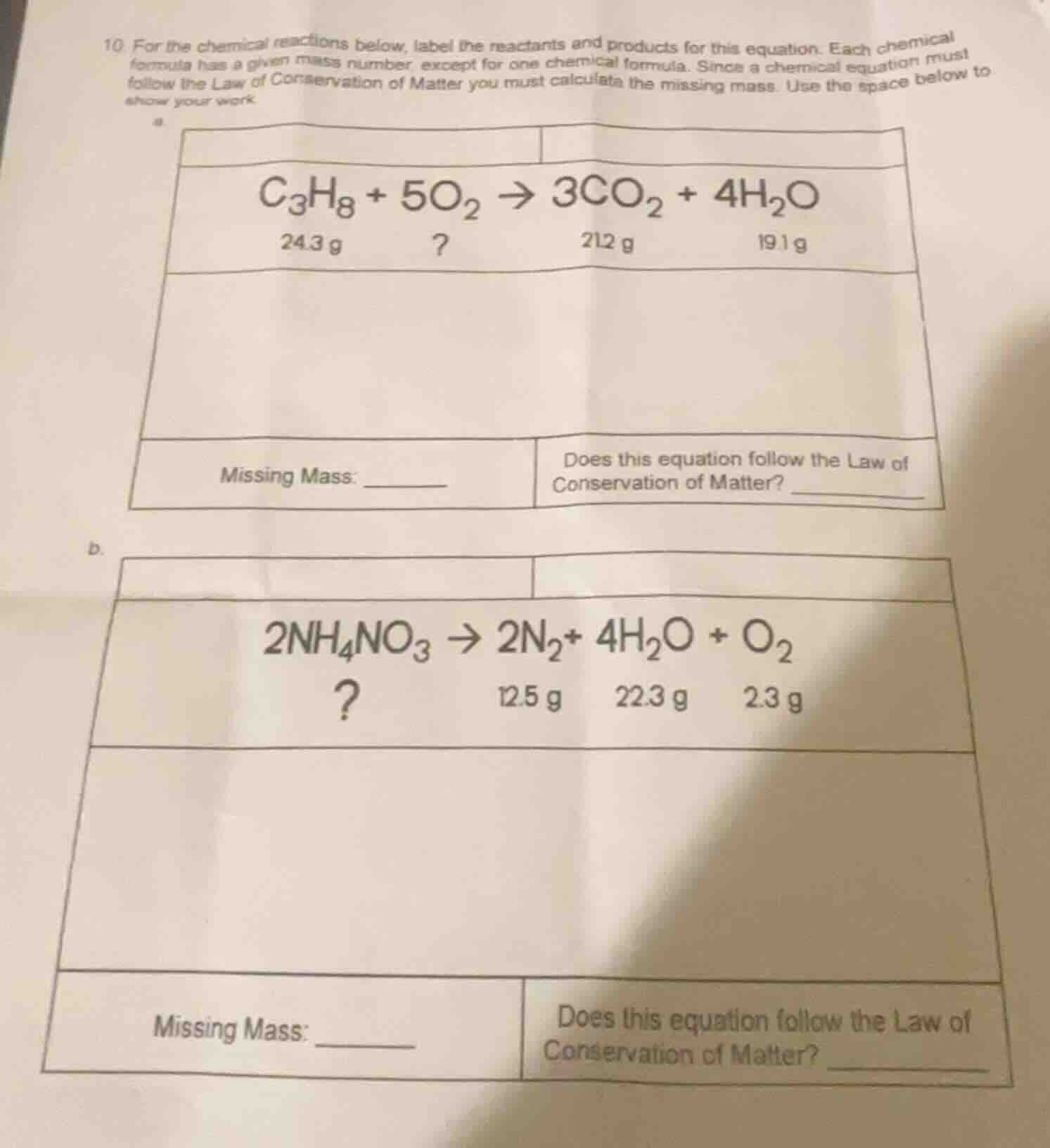
- for the chemical reactions below, label the reactants and products for this equation. each chemical formula has a given mass number, except for one chemical formula. since a chemical equation must follow the law of conservation of matter you must calculate the missing mass. use the space below to show your work.
a.
| |
| $\ce{c_{3}h_{8} + 5o_{2} \
ightarrow 3co_{2} + 4h_{2}o}$ |
| 24.3 g ? 212 g 19.1 g |
| missing mass: ______ | does this equation follow the law of conservation of matter? ______ |
b.
| |
| $\ce{2nh_{4}no_{3} \
ightarrow 2n_{2} + 4h_{2}o + o_{2}}$ |
| ? 12.5 g 22.3 g 2.3 g |
| missing mass: ______ | does this equation follow the law of conservation of matter? ______ |
Part a
Step1: Calculate total product mass
Add masses of $\text{CO}_2$ and $\text{H}_2\text{O}$:
$212\ \text{g} + 19.1\ \text{g} = 231.1\ \text{g}$
Step2: Find missing reactant mass
Subtract mass of $\text{C}_3\text{H}_8$ from total product mass:
$231.1\ \text{g} - 24.3\ \text{g} = 206.8\ \text{g}$
Step3: Verify mass conservation
Total reactant mass: $24.3\ \text{g} + 206.8\ \text{g} = 231.1\ \text{g}$
Total product mass: $231.1\ \text{g}$
Masses are equal, so the law holds.
Part b
Step1: Calculate total product mass
Add masses of $\text{N}_2$, $\text{H}_2\text{O}$, and $\text{O}_2$:
$12.5\ \text{g} + 22.3\ \text{g} + 2.3\ \text{g} = 37.1\ \text{g}$
Step2: Identify missing reactant mass
Per conservation of matter, total reactant mass equals total product mass, so missing mass is $37.1\ \text{g}$.
Step3: Verify mass conservation
Total reactant mass = $37.1\ \text{g}$, total product mass = $37.1\ \text{g}$
Masses are equal, so the law holds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part a
Missing Mass: $206.8\ \text{g}$
Does this equation follow the Law of Conservation of Matter? Yes
Part b
Missing Mass: $37.1\ \text{g}$
Does this equation follow the Law of Conservation of Matter? Yes