QUESTION IMAGE
Question
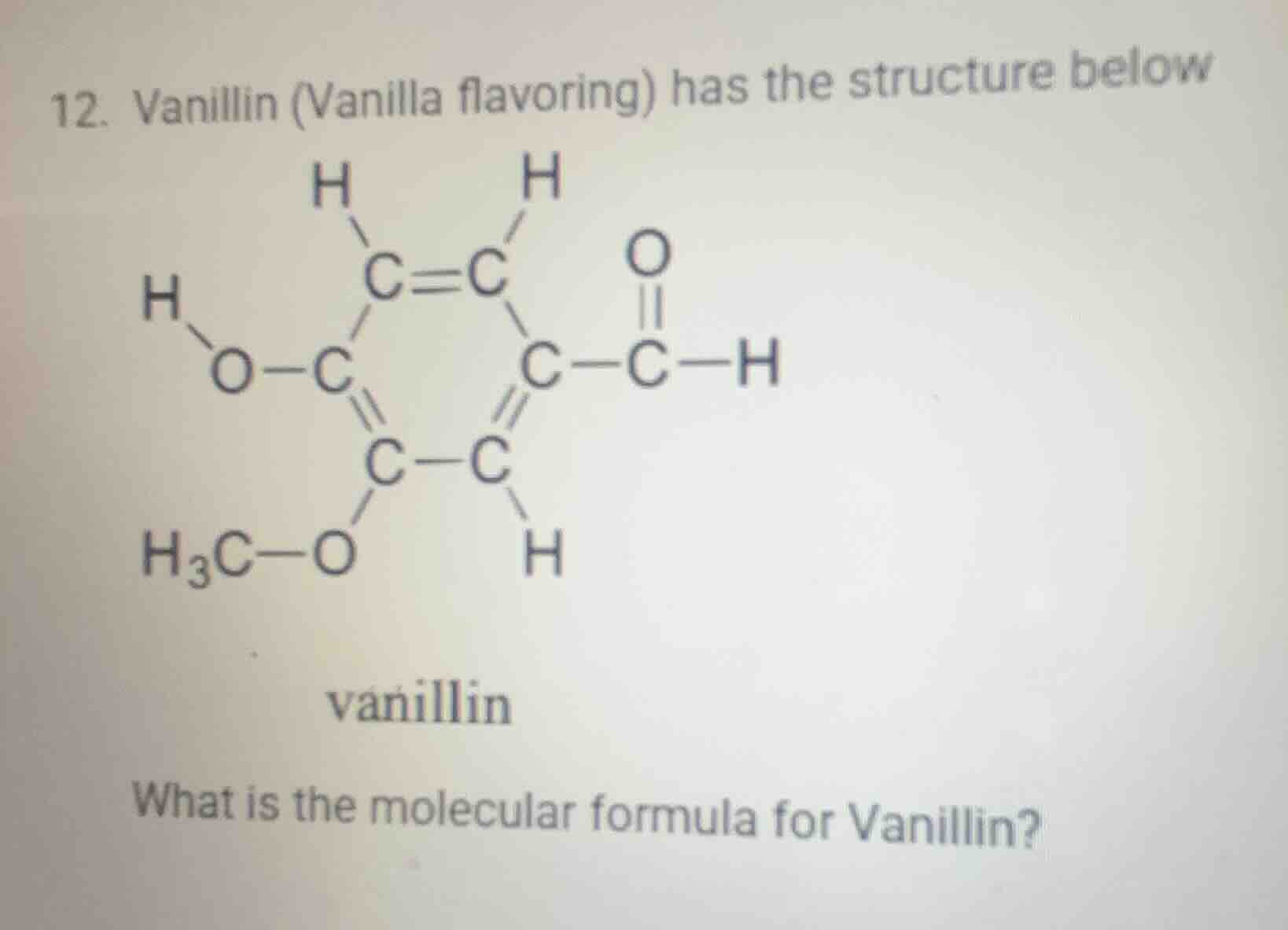
- vanillin (vanilla flavoring) has the structure below
vanillin
what is the molecular formula for vanillin?
Step1: Count Carbon atoms
Looking at the structure, we identify each carbon: the ring has 6 carbons, and the carbonyl - attached carbon, so total C: 8? Wait, no, let's re - examine. Wait, the benzene - like ring? Wait, no, let's count each C atom:
- The double - bonded C (C = C) two, then the C connected to O - H, the C connected to O - CH₃, the C in between, the C connected to the carbonyl group, and the ring? Wait, maybe a better way: let's list each C:
- C with H and double bond (C = C)
- C with H and double bond (C = C)
- C connected to O - H (single bond to O - H, double bond to another C)
- C connected to O - CH₃ (single bond to O - CH₃, double bond to another C)
- C between 3 and 4 (single bond)
- C between 2 and the carbonyl - C (single bond)
- The carbonyl - C (double bond to O, single bond to H and to C - 6)
Wait, no, I think I made a mistake. Let's count again. Let's look at the structure:
- The aromatic - like ring (but with some double bonds): Let's count C atoms:
First, the ring: let's see, the structure has a 6 - membered ring? Wait, no, the bonds:
Looking at the structure:
- C = C (two Hs)
- C connected to that C (double bond to another C, single bond to the carbonyl - C)
- Carbonyl - C (double bond to O, single bond to H and to the previous C)
- C in the ring: let's count all C:
Wait, maybe a standard way: Vanillin's molecular formula is known, but let's count from the structure.
Count C: Let's see, the ring has 6 carbons? No, wait, the structure:
- The C with H - O: 1
- The C with H₃C - O: 1
- The C between them: 1
- The C between the two C = C: 1
- The two C = C: 2
- The C connected to the carbonyl (C = O): 1
Wait, that's 1 + 1+1 + 1+2 + 1=7? No, no, I think I messed up. Wait, let's do it properly.
Looking at the structure:
- Let's label each C:
- C (H, double bond to C2)
- C (H, double bond to C1, single bond to C6)
- C (single bond to O - H, double bond to C4)
- C (single bond to O - CH₃, double bond to C3)
- C (single bond to C3 and C4)
- C (single bond to C2 and C7)
- C (double bond to O, single bond to H and C6)
Now count C: 7? No, that can't be. Wait, Vanillin's actual formula is C₈H₈O₃. Wait, maybe I missed a C. Oh, wait, the O - CH₃ has a C (H₃C - O), so that's a C. So:
- C in H₃C - O: 1
- C in O - H: 1
- The two C = C: 2
- The C between O - H and O - CH₃: 1
- The C between C = C and carbonyl - C: 1
- The carbonyl - C: 1
- The C in the middle of the two C = C? No, wait, maybe the ring is 6 - membered? Wait, no, let's count H atoms.
Count H atoms:
- On C = C: 2 Hs (each C = C has one H? Wait, no, the first C = C: one H, second C = C: one H? Wait, the structure shows H - C = C - H, so two Hs (one on each C of the double bond). Then:
- H on O - H: 1
- H on the carbonyl - C: 1
- H on the C connected to H (the single - bonded H on the ring): 1
- H₃C: 3 Hs
Wait, let's count H:
- From H - O: 1
- From H₃C: 3
- From H - C = C - H: 2
- From the single - bonded H on the ring (the C with H): 1
- From the carbonyl - H: 1
- Any other H? Wait, maybe the ring has some Hs? Wait, no, let's do it step by step.
Count C:
- H₃C: 1 C
- O - H connected C: 1 C
- Two C = C: 2 C
- The C between O - H and O - CH₃: 1 C
- The C between C = C and carbonyl - C: 1 C
- The carbonyl - C: 1 C
Wait, that's 1+1 + 2+1+1+1 = 7? No, that's wrong. Wait, Vanillin's formula is C₈H₈O₃. Ah, I see, I missed a C. Wait, the carbonyl - C is connected to a C in the ring, and the ring has 6 carbons? Wait, maybe the ring is 6 - membered, and then the carbonyl - C is an additional C. So ring C: 6, carbonyl - C:1, and th…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The molecular formula of Vanillin is $\boldsymbol{C_8H_8O_3}$