QUESTION IMAGE
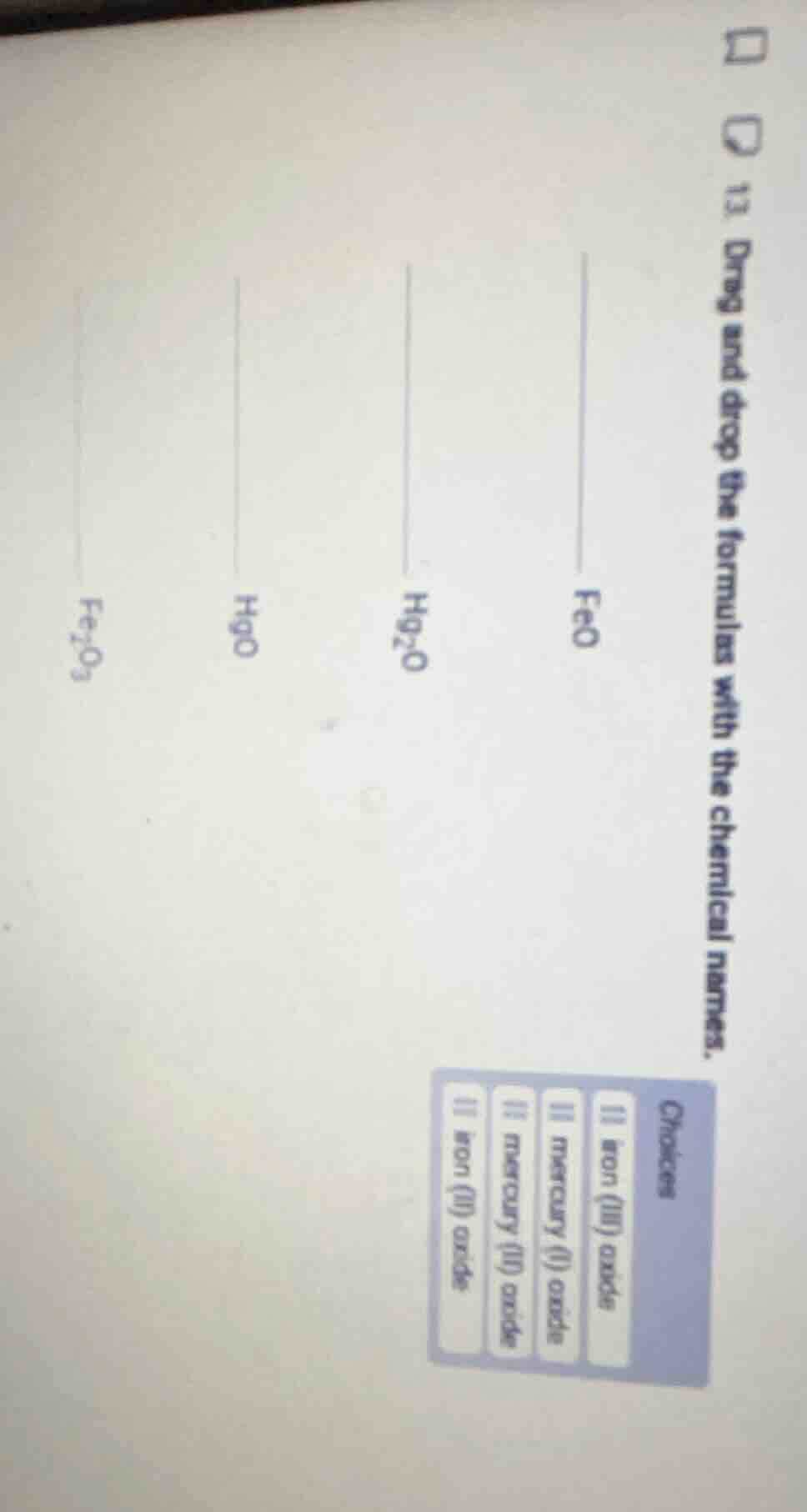
Question
- drag and drop the formulas with the chemical names.
formulas: feo, hg₂o, hgo, fe₂o₃
choices:
- iron (iii) oxide
- mercury (i) oxide
- mercury (ii) oxide
- iron (ii) oxide
Brief Explanations
Match each formula to its name by balancing ionic charges:
- For FeO: Iron has a +2 charge, oxide is -2, so it is iron (II) oxide.
- For $\text{Hg}_2\text{O}$: Mercury has a +1 charge (two Hg⁺ balance one O²⁻), so it is mercury (I) oxide.
- For HgO: Mercury has a +2 charge, oxide is -2, so it is mercury (II) oxide.
- For $\text{Fe}_2\text{O}_3$: Iron has a +3 charge (two Fe³⁺ balance three O²⁻), so it is iron (III) oxide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{FeO}$: iron (II) oxide
- $\text{Hg}_2\text{O}$: mercury (I) oxide
- $\text{HgO}$: mercury (II) oxide
- $\text{Fe}_2\text{O}_3$: iron (III) oxide