QUESTION IMAGE
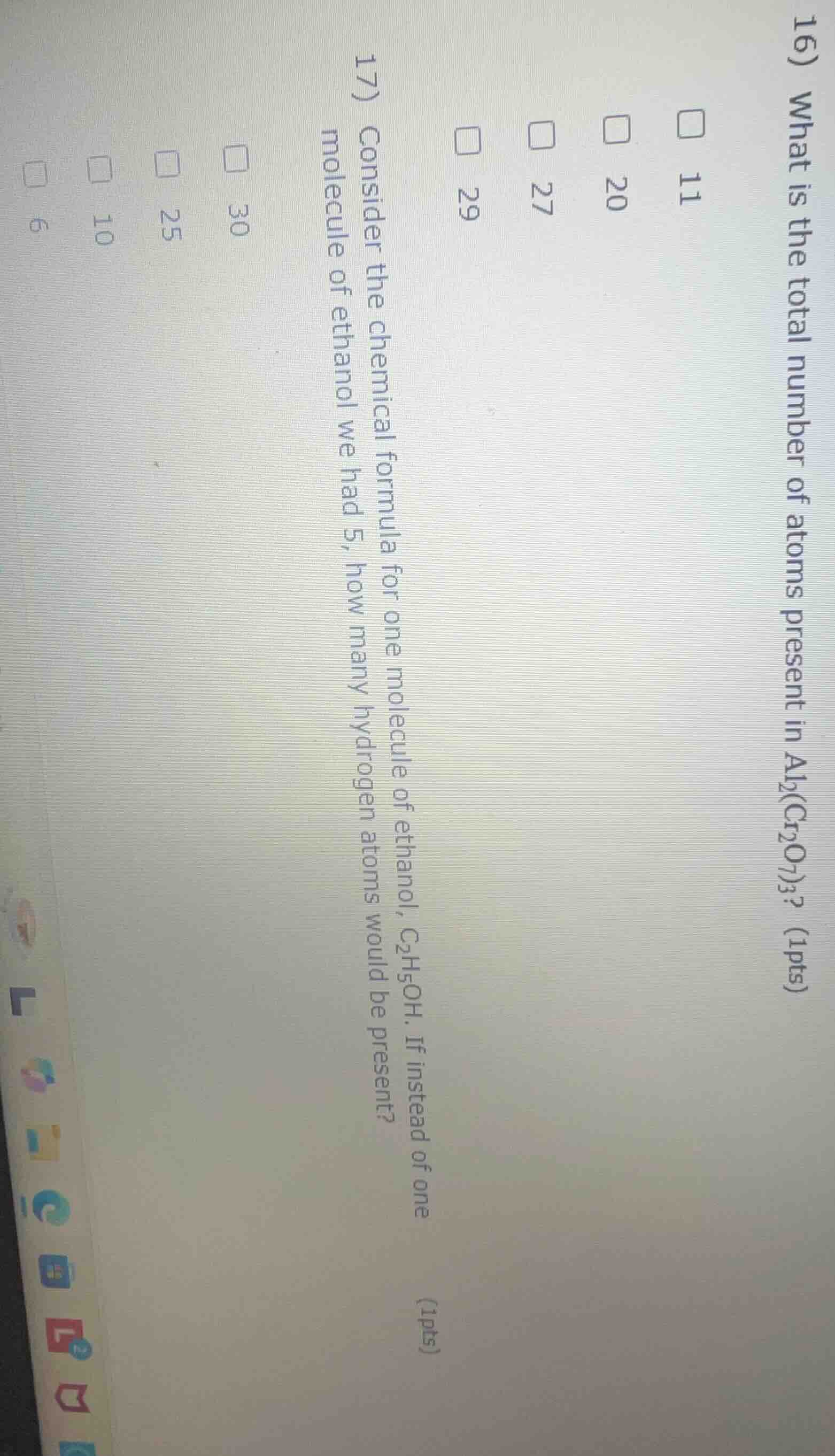
Question
- consider the chemical formula for one molecule of ethanol, c₂h₅oh. if instead of one molecule of ethanol we had 5, how many hydrogen atoms would be present? (1pts) 30 25 10 6 16) what is the total number of atoms present in al₂(cr₂o₇)₃? (1pts) 11 20 27 29
Step1: Count atoms in $\text{Al}_2(\text{Cr}_2\text{O}_7)_3$
Step1a: Calculate Al atoms
$2$ (from subscript of Al)
Step1b: Calculate Cr atoms
$2 \times 3 = 6$
Step1c: Calculate O atoms
$7 \times 3 = 21$
Step1d: Sum all atoms
$2 + 6 + 21 = 29$
Step2: Calculate H atoms in 5 ethanol
Step2a: H per ethanol molecule
$5 + 1 = 6$ (from $\text{C}_2\text{H}_5\text{OH}$)
Step2b: Total H atoms
$6 \times 5 = 30$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{29}$
- $\boldsymbol{30}$