QUESTION IMAGE
Question
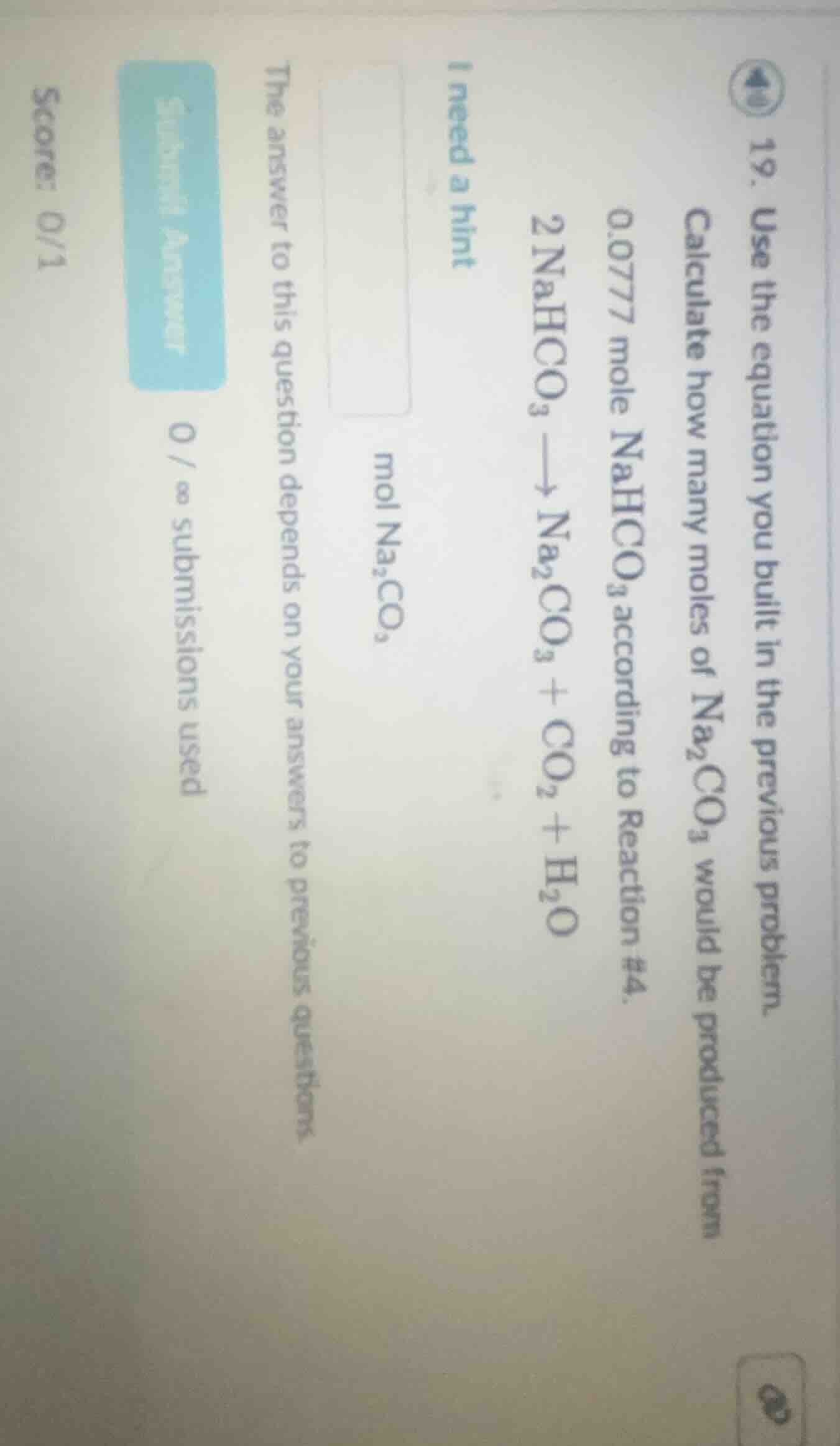
- use the equation you built in the previous problem.
calculate how many moles of $ce{na_{2}co_{3}}$ would be produced from 0.0777 mole $ce{nahco_{3}}$ according to reaction #4.
$ce{2nahco_{3} -> na_{2}co_{3} + co_{2} + h_{2}o}$
i need a hint
$\boldsymbol{ce{mol na_{2}co_{3}}}$
the answer to this question depends on your answers to previous questions.
submit answer
0 / ∞ submissions used
score: 0/1
Step1: Identify mole ratio
From the balanced equation $2\text{NaHCO}_3
ightarrow \text{Na}_2\text{CO}_3 + \text{CO}_2 + \text{H}_2\text{O}$, the mole ratio of $\text{NaHCO}_3$ to $\text{Na}_2\text{CO}_3$ is $2:1$.
Step2: Calculate moles of $\text{Na}_2\text{CO}_3$
Moles of $\text{Na}_2\text{CO}_3 = \frac{\text{Moles of NaHCO}_3}{2}$
$\text{Moles of Na}_2\text{CO}_3 = \frac{0.0777}{2}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.03885 mol