QUESTION IMAGE
Question
22 mark for review
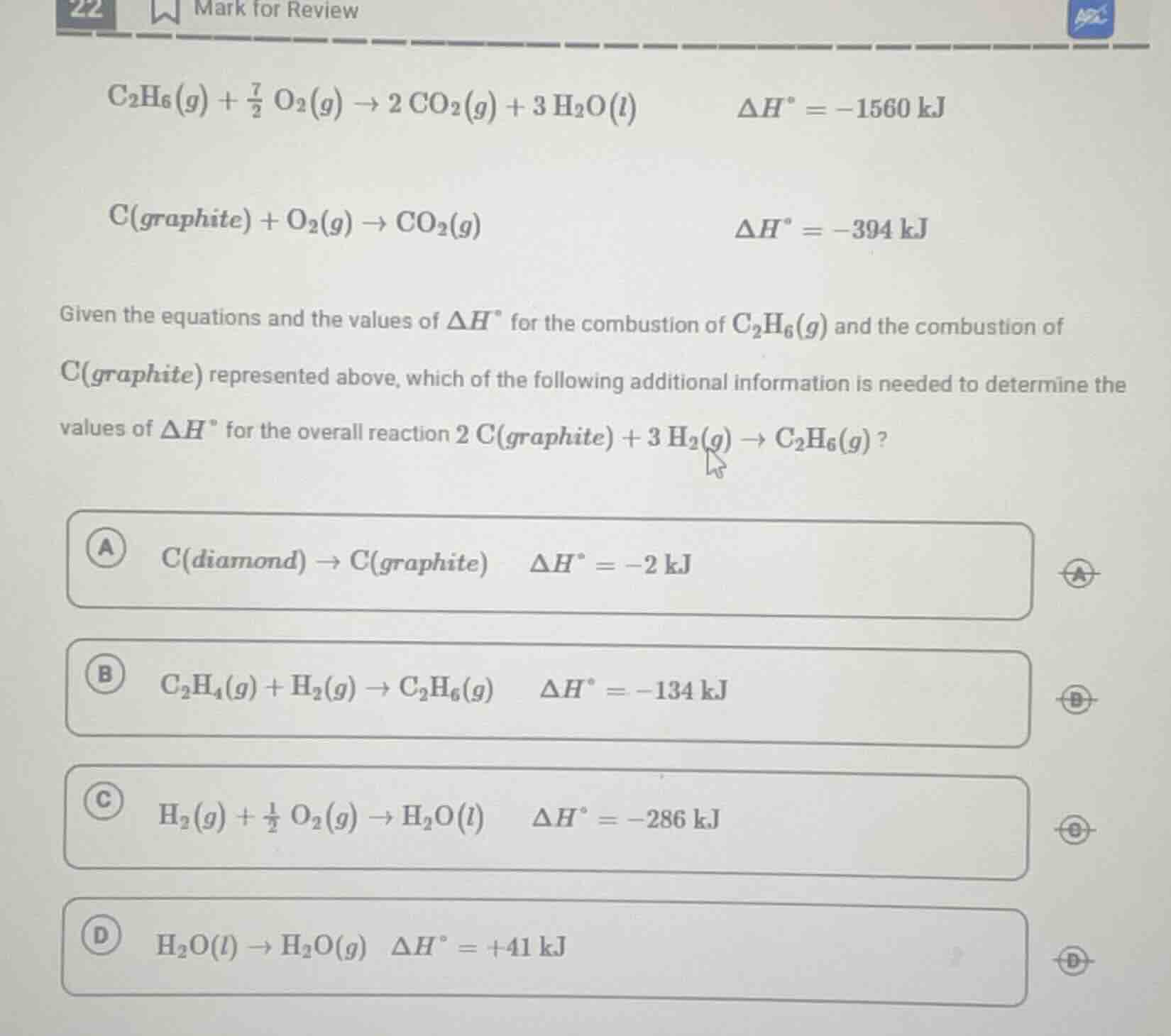
\\(\ce{c_{2}h_{6}(g) + \frac{7}{2} o_{2}(g) -> 2 co_{2}(g) + 3 h_{2}o(l)} \quad \delta h^\circ = -1560 \\, \text{kj}\\)
\\(\ce{c(graphite) + o_{2}(g) -> co_{2}(g)} \quad \delta h^\circ = -394 \\, \text{kj}\\)
given the equations and the values of \\(\delta h^\circ\\) for the combustion of \\(\ce{c_{2}h_{6}(g)}\\) and the combustion of \\(\ce{c(graphite)}\\) represented above, which of the following additional information is needed to determine the values of \\(\delta h^\circ\\) for the overall reaction \\(\ce{2 c(graphite) + 3 h_{2}(g) -> c_{2}h_{6}(g)}\\)?
a \\(\ce{c(diamond) -> c(graphite)} \quad \delta h^\circ = -2 \\, \text{kj}\\)
b \\(\ce{c_{2}h_{4}(g) + h_{2}(g) -> c_{2}h_{6}(g)} \quad \delta h^\circ = -134 \\, \text{kj}\\)
c \\(\ce{h_{2}(g) + \frac{1}{2} o_{2}(g) -> h_{2}o(l)} \quad \delta h^\circ = -286 \\, \text{kj}\\)
d \\(\ce{h_{2}o(l) -> h_{2}o(g)} \quad \delta h^\circ = +41 \\, \text{kj}\\)
To determine the enthalpy change ($\Delta H^{\circ}$) for the reaction $2\ \text{C(graphite)} + 3\ \text{H}_2(g)
ightarrow \text{C}_2\text{H}_6(g)$, we can use Hess's Law. Let's analyze the given reactions and what we need:
- The combustion of $\text{C}_2\text{H}_6$: $\text{C}_2\text{H}_6(g) + \frac{7}{2}\ \text{O}_2(g)
ightarrow 2\ \text{CO}_2(g) + 3\ \text{H}_2\text{O}(l)$ with $\Delta H^{\circ} = -1560\ \text{kJ}$. If we reverse this reaction, we get $2\ \text{CO}_2(g) + 3\ \text{H}_2\text{O}(l)
ightarrow \text{C}_2\text{H}_6(g) + \frac{7}{2}\ \text{O}_2(g)$ with $\Delta H^{\circ} = +1560\ \text{kJ}$.
- The combustion of $\text{C(graphite)}$: $\text{C(graphite)} + \text{O}_2(g)
ightarrow \text{CO}_2(g)$ with $\Delta H^{\circ} = -394\ \text{kJ}$. If we multiply this reaction by 2, we get $2\ \text{C(graphite)} + 2\ \text{O}_2(g)
ightarrow 2\ \text{CO}_2(g)$ with $\Delta H^{\circ} = 2 \times (-394\ \text{kJ}) = -788\ \text{kJ}$.
Now, let's add the reversed $\text{C}_2\text{H}_6$ combustion and the multiplied $\text{C(graphite)}$ combustion:
$2\ \text{CO}_2(g) + 3\ \text{H}_2\text{O}(l) + 2\ \text{C(graphite)} + 2\ \text{O}_2(g)
ightarrow \text{C}_2\text{H}_6(g) + \frac{7}{2}\ \text{O}_2(g) + 2\ \text{CO}_2(g)$
Simplifying (cancel $2\ \text{CO}_2(g)$ and subtract $2\ \text{O}_2(g)$ from $\frac{7}{2}\ \text{O}_2(g)$ to get $\frac{3}{2}\ \text{O}_2(g)$), we have:
$2\ \text{C(graphite)} + 3\ \text{H}_2\text{O}(l)
ightarrow \text{C}_2\text{H}_6(g) + \frac{3}{2}\ \text{O}_2(g)$ with $\Delta H^{\circ} = +1560\ \text{kJ} - 788\ \text{kJ} = +772\ \text{kJ}$.
To get to the target reaction $2\ \text{C(graphite)} + 3\ \text{H}_2(g)
ightarrow \text{C}_2\text{H}_6(g)$, we need to replace $3\ \text{H}_2\text{O}(l)$ and $\frac{3}{2}\ \text{O}_2(g)$ with $3\ \text{H}_2(g)$. This means we need the enthalpy change for the reaction of $\text{H}_2(g)$ with $\text{O}_2(g)$ to form $\text{H}_2\text{O}(l)$, i.e., the combustion of $\text{H}_2$: $\text{H}_2(g) + \frac{1}{2}\ \text{O}_2(g)
ightarrow \text{H}_2\text{O}(l)$. If we multiply this by 3, we get $3\ \text{H}_2(g) + \frac{3}{2}\ \text{O}_2(g)
ightarrow 3\ \text{H}_2\text{O}(l)$ with $\Delta H^{\circ} = 3 \times (-286\ \text{kJ})$ (if we know the $\Delta H^{\circ}$ for $\text{H}_2(g) + \frac{1}{2}\ \text{O}_2(g)
ightarrow \text{H}_2\text{O}(l)$).
Let's check the options:
- Option A: The reaction between diamond and graphite is irrelevant here, as we are dealing with graphite and $\text{H}_2$. Eliminate A.
- Option B: The reaction involving $\text{C}_2\text{H}_4$ is not related to the formation of $\text{C}_2\text{H}_6$ from graphite and $\text{H}_2$ in terms of the needed steps. Eliminate B.
- Option C: This is the combustion of $\text{H}_2$ to form $\text{H}_2\text{O}(l)$, which is exactly what we need. If we have this, we can multiply it by 3 and add to the previous simplified reaction to get the target reaction.
- Option D: The phase change of $\text{H}_2\text{O}$ (liquid to gas) is not needed, as our reactions involve $\text{H}_2\text{O}(l)$. Eliminate D.
So, the additional information needed is the enthalpy change for the combustion of $\text{H}_2$ to form $\text{H}_2\text{O}(l)$, which is given in Option C.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. $\text{H}_2(g) + \frac{1}{2}\ \text{O}_2(g)
ightarrow \text{H}_2\text{O}(l)$ $\Delta H^{\circ} = -286\ \text{kJ}$