QUESTION IMAGE
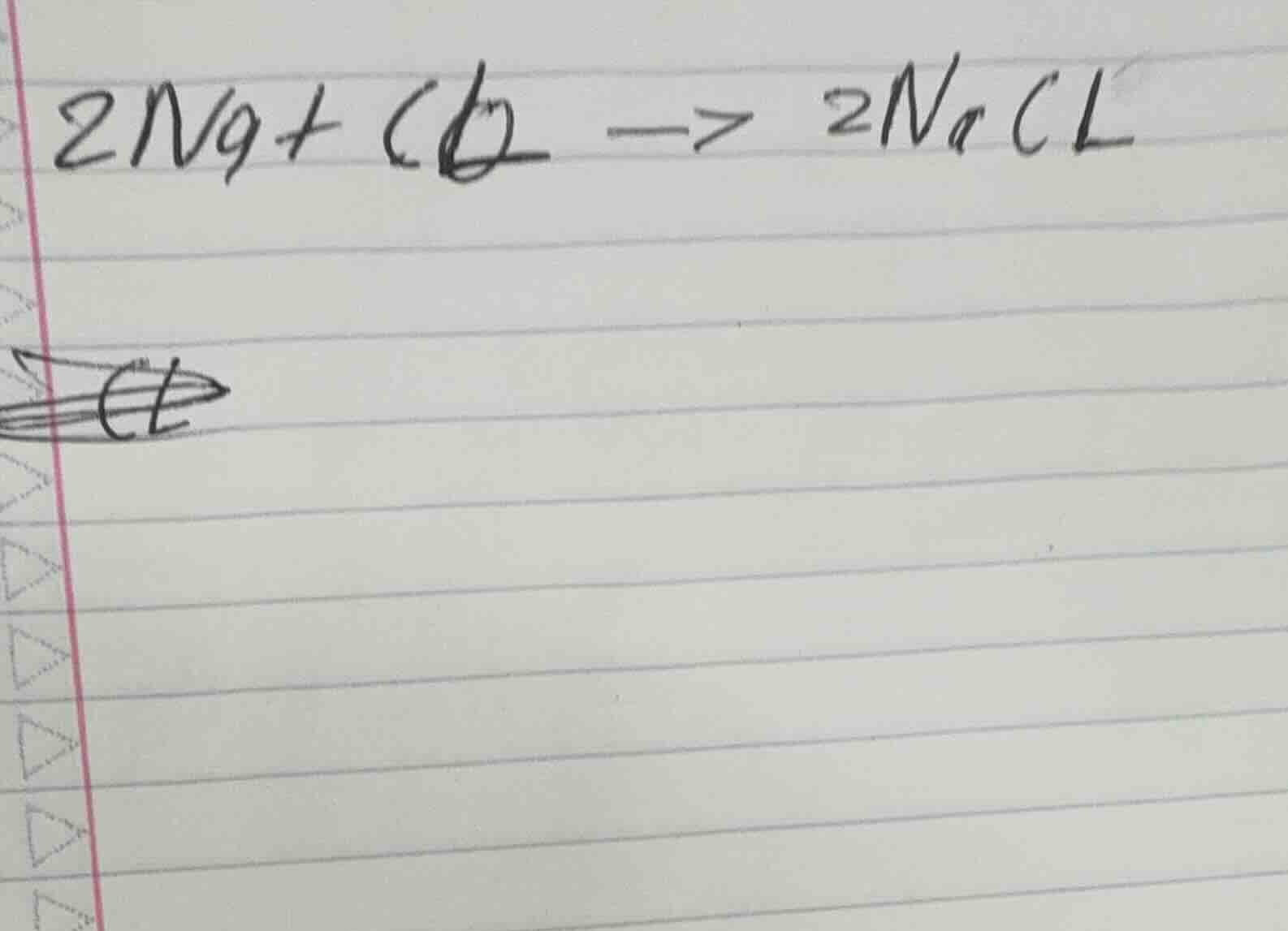
Question
2ng + cl₂ -> 2nacl
Step1: Correct chemical symbols
First, fix miswritten symbols: $Na$ (not $Ng$), $\text{Cl}_2$ (not $Cl$).
Step2: Balance and format reaction
Verify atom counts: 2 Na, 2 Cl on both sides. Add reaction condition (heat, $\Delta$) and correct arrow.
$$2\text{Na} + \text{Cl}_2 \xrightarrow{\Delta} 2\text{NaCl}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The corrected, balanced chemical equation is:
$$2\text{Na} + \text{Cl}_2 \xrightarrow{\Delta} 2\text{NaCl}$$
(Note: The original had typographical errors in element symbols; sodium is $\text{Na}$, chlorine gas is diatomic $\text{Cl}_2$, and the reaction requires heat, indicated by $\Delta$ above the arrow.)