QUESTION IMAGE
Question
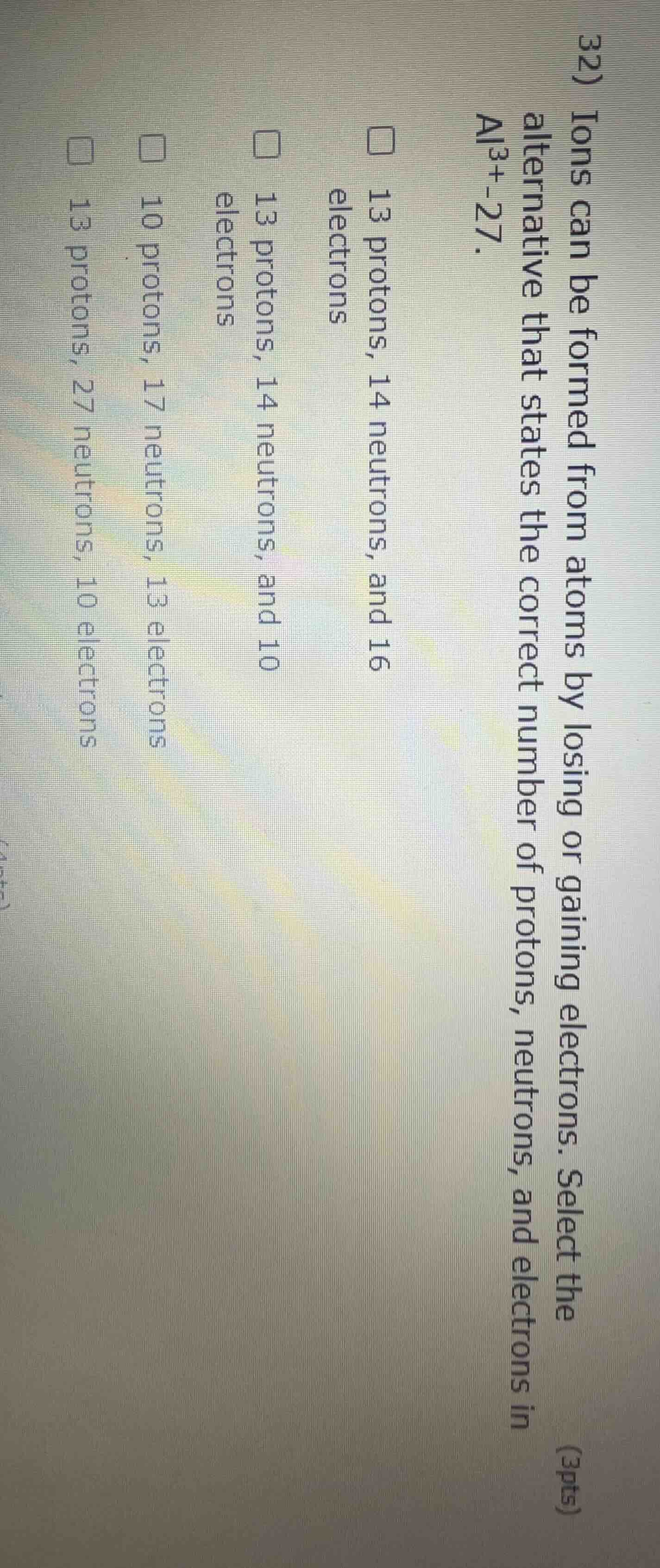
- ions can be formed from atoms by losing or gaining electrons. select the alternative that states the correct number of protons, neutrons, and electrons in al³⁺-27. 13 protons, 14 neutrons, and 16 electrons 13 protons, 14 neutrons, and 10 electrons 10 protons, 17 neutrons, 13 electrons 13 protons, 27 neutrons, 10 electrons
Step1: Find number of protons
The atomic number of Al is 13, so protons = 13.
Step2: Calculate number of neutrons
Mass number = 27, neutrons = $27 - 13 = 14$.
Step3: Calculate number of electrons
Al$^{3+}$ loses 3 electrons; neutral Al has 13 electrons, so electrons = $13 - 3 = 10$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
13 protons, 14 neutrons, and 10 electrons