QUESTION IMAGE
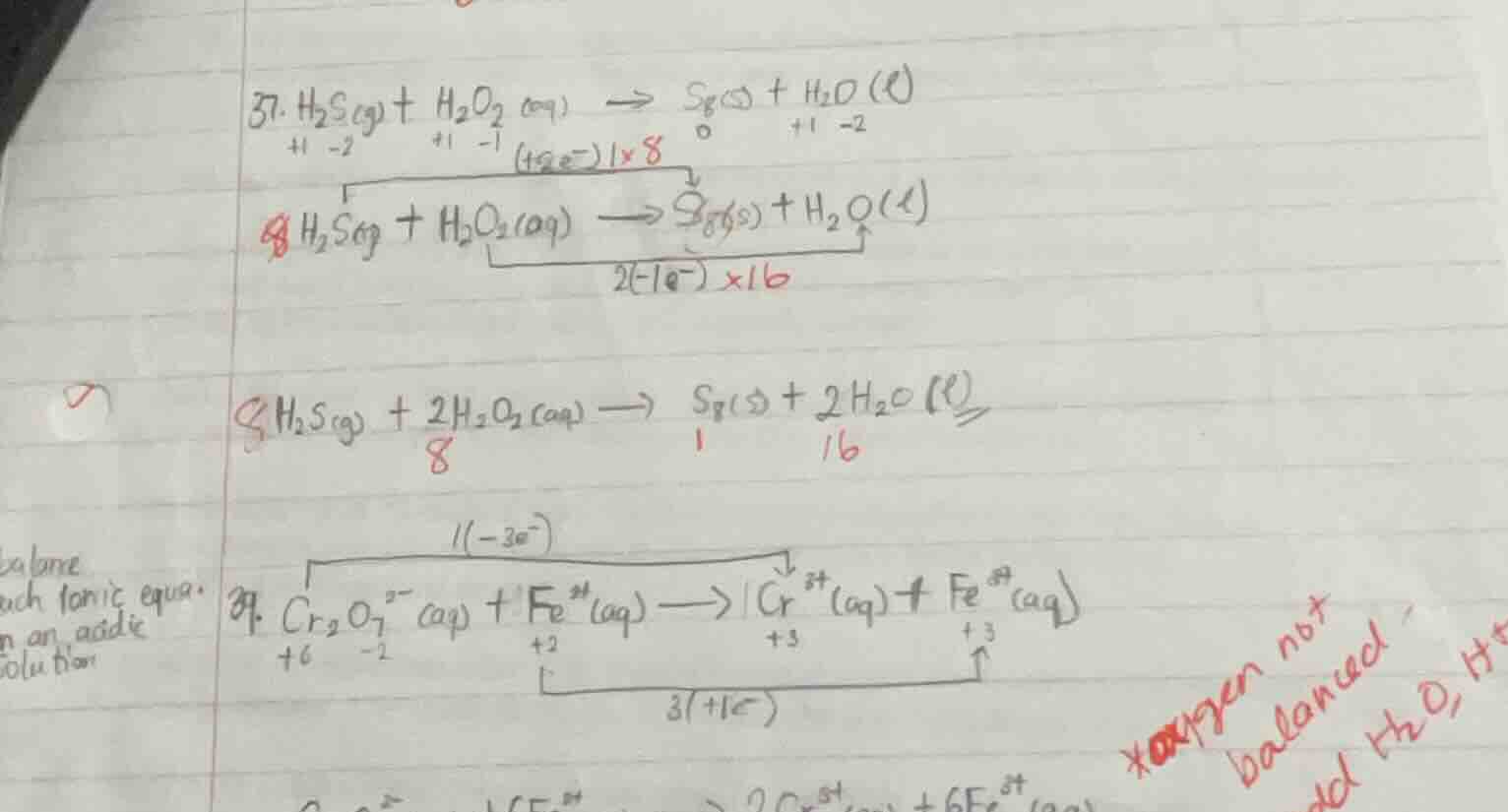
Question
- \\( \ce{h2s(g) + h2o2(aq) -> s8(s) + h2o(l)} \\)
\\( \ce{h2s(g) + h2o2(aq) -> s8(s) + h2o(l)} \\)
\\( \ce{h2s(g) + 2h2o2(aq) -> s8(s) + 2h2o(l)} \\)
- \\( \ce{cr2o7^{2-}(aq) + fe^{2+}(aq) -> cr^{3+}(aq) + fe^{3+}(aq)} \\)
balance
each ionic equa.
in an acidic
solution
Step 1: Balance the sulfur atoms
In the reaction \( \ce{H2S(g) + H2O2(aq) -> S8(s) + H2O(l)} \), there are 8 S atoms in \( \ce{S8} \), so we need 8 \( \ce{H2S} \) molecules. So the equation becomes \( \ce{8H2S(g) + H2O2(aq) -> S8(s) + H2O(l)} \).
Step 2: Balance the oxygen and hydrogen atoms (using redox method or simple balancing)
Now, let's balance the oxygen and hydrogen. Let's look at the oxidation states. S in \( \ce{H2S} \) is -2, in \( \ce{S8} \) is 0 (oxidation, loses 2e⁻ per S atom, 8 S atoms lose \( 8\times2 = 16 \) e⁻). O in \( \ce{H2O2} \) is -1, in \( \ce{H2O} \) is -2 (reduction, gains 1e⁻ per O atom). To balance electrons, the number of electrons gained should equal electrons lost. So for O: each \( \ce{H2O2} \) has 2 O atoms, gains 2e⁻ per \( \ce{H2O2} \) molecule. To gain 16e⁻, we need \( \frac{16}{2}=8 \) \( \ce{H2O2} \) molecules? Wait, no, earlier step had a mistake. Wait, let's do simple balancing.
Wait, original unbalanced: \( \ce{H2S + H2O2 -> S8 + H2O} \)
Balance S: 8 \( \ce{H2S} \) gives 1 \( \ce{S8} \): \( \ce{8H2S + H2O2 -> S8 + H2O} \)
Now balance H: left side has \( 8\times2 + 2 = 18 \) H? No, \( 8\ce{H2S} \) has \( 16 \) H, \( \ce{H2O2} \) has 2 H, total 18 H. Right side \( \ce{H2O} \) has 2 H per molecule. Let \( x \) be the number of \( \ce{H2O} \) molecules. So \( 2x = 18 \)? No, that's not right. Wait, maybe the redox approach is better.
Oxidation: \( \ce{H2S -> S8} \), S from -2 to 0. 8 S atoms: \( 8\ce{H2S -> S8 + 16H+ + 16e-} \) (since each \( \ce{H2S} \) gives \( \ce{S} \), loses 2e⁻, 8 \( \ce{H2S} \) lose 16e⁻)
Reduction: \( \ce{H2O2 -> H2O} \), O from -1 to -2. Each \( \ce{H2O2} \) has 2 O atoms, gains 2e⁻ (since each O gains 1e⁻, 2 O atoms gain 2e⁻ per \( \ce{H2O2} \)). So reduction half-reaction: \( \ce{H2O2 + 2H+ + 2e- -> 2H2O} \) (wait, balancing O and H: \( \ce{H2O2 -> H2O} \), add 1 \( \ce{H2O} \) on left? No, better: \( \ce{H2O2 + 2H+ + 2e- -> 2H2O} \) (now O: 2 on left, 2 on right; H: 4 on left (2 from \( \ce{H2O2} \), 2 from \( \ce{H+} \)), 4 on right (2 \( \ce{H2O} \))).
Now, electrons lost: 16e⁻ (from oxidation), electrons gained: 2e⁻ per reduction half-reaction. So multiply reduction half-reaction by 8: \( \ce{8H2O2 + 16H+ + 16e- -> 16H2O} \)
Now add oxidation and reduction half-reactions:
Oxidation: \( \ce{8H2S -> S8 + 16H+ + 16e-} \)
Reduction: \( \ce{8H2O2 + 16H+ + 16e- -> 16H2O} \)
Total: \( \ce{8H2S + 8H2O2 -> S8 + 16H2O} \)
Simplify: divide all coefficients by... Wait, no, let's check. 8 \( \ce{H2S} \), 8 \( \ce{H2O2} \), 1 \( \ce{S8} \), 16 \( \ce{H2O} \). Let's check H: 82 + 82 = 16 + 16 = 32; right side 162 = 32. O: 82 = 16; right side 161 = 16. S: 81 = 8; right side 8. So balanced equation is \( \ce{8H2S(g) + 8H2O2(aq) -> S8(s) + 16H2O(l)} \) or we can divide by 8? Wait, no, original problem's handwritten had a mistake. Wait, the correct balanced equation for \( \ce{H2S + H2O2 -> S8 + H2O} \) is:
\( \ce{8H2S + 8H2O2 -> S8 + 16H2O} \) (dividing by 8: \( \ce{H2S + H2O2 -> \frac{1}{8}S8 + 2H2O} \), no, better to keep as \( \ce{8H2S + 8H2O2 -> S8 + 16H2O} \))
Wait, but the handwritten had \( \ce{8H2S + 16H2O2 -> S8 + 16H2O} \)? No, let's do it again.
Wait, S: 8 \( \ce{H2S} \) → 1 \( \ce{S8} \)
O: each \( \ce{H2O2} \) has 2 O, each \( \ce{H2O} \) has 1 O. Let number of \( \ce{H2O2} \) be x, \( \ce{H2O} \) be y.
Then 2x = y (O balance)
H balance: 2*8 + 2x = 2y (since \( \ce{H2S} \) has 2 H, \( \ce{H2O2} \) has 2 H, \( \ce{H2O} \) has 2 H)
Substitute y = 2x into H balance: 16 + 2x = 2*(2x) → 16 + 2x = 4x → 16 = 2x → x = 8, then y = 16.
So equation:…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The balanced chemical equation is \( \boldsymbol{\ce{8H2S(g) + 8H2O2(aq) -> S8(s) + 16H2O(l)}} \) (or can be simplified by dividing all coefficients by 8: \( \ce{H2S(g) + H2O2(aq) -> \frac{1}{8}S8(s) + 2H2O(l)} \), but usually we use whole numbers, so the first one is better with whole coefficients).