QUESTION IMAGE
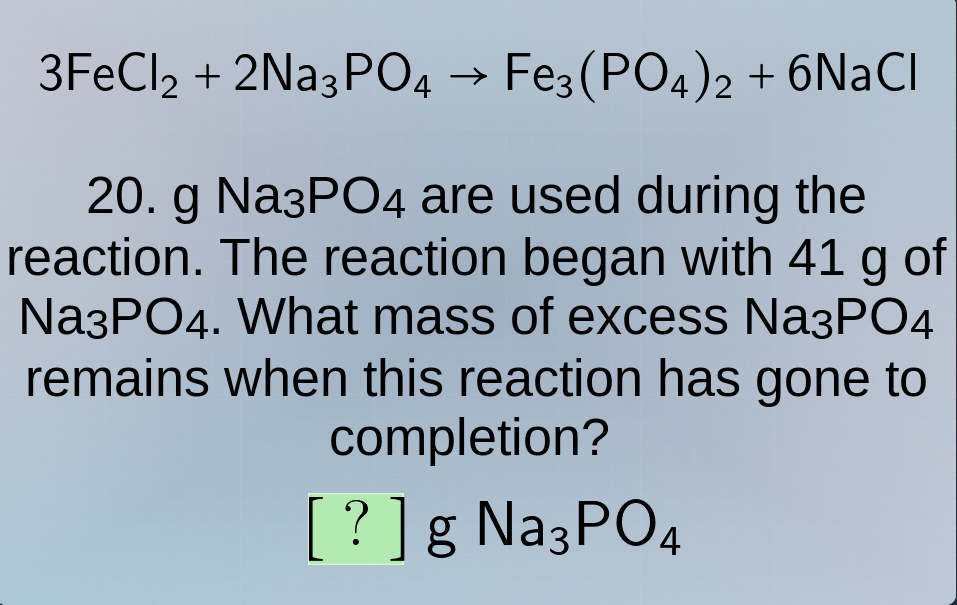
Question
3fecl₂ + 2na₃po₄ → fe₃(po₄)₂ + 6nacl
- g na₃po₄ are used during the reaction. the reaction began with 41 g of na₃po₄. what mass of excess na₃po₄ remains when this reaction has gone to completion?
? g na₃po₄
Step1: Identify the given amounts
We know the initial mass of $\ce{Na3PO4}$ is $41\ \text{g}$ and the mass of $\ce{Na3PO4}$ used in the reaction is $20\ \text{g}$.
Step2: Calculate the excess mass
To find the mass of excess $\ce{Na3PO4}$, we subtract the mass used from the initial mass. The formula for excess mass ($m_{\text{excess}}$) is:
$$m_{\text{excess}} = m_{\text{initial}} - m_{\text{used}}$$
Substituting the given values:
$$m_{\text{excess}} = 41\ \text{g} - 20\ \text{g}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$21\ \text{g}$