QUESTION IMAGE
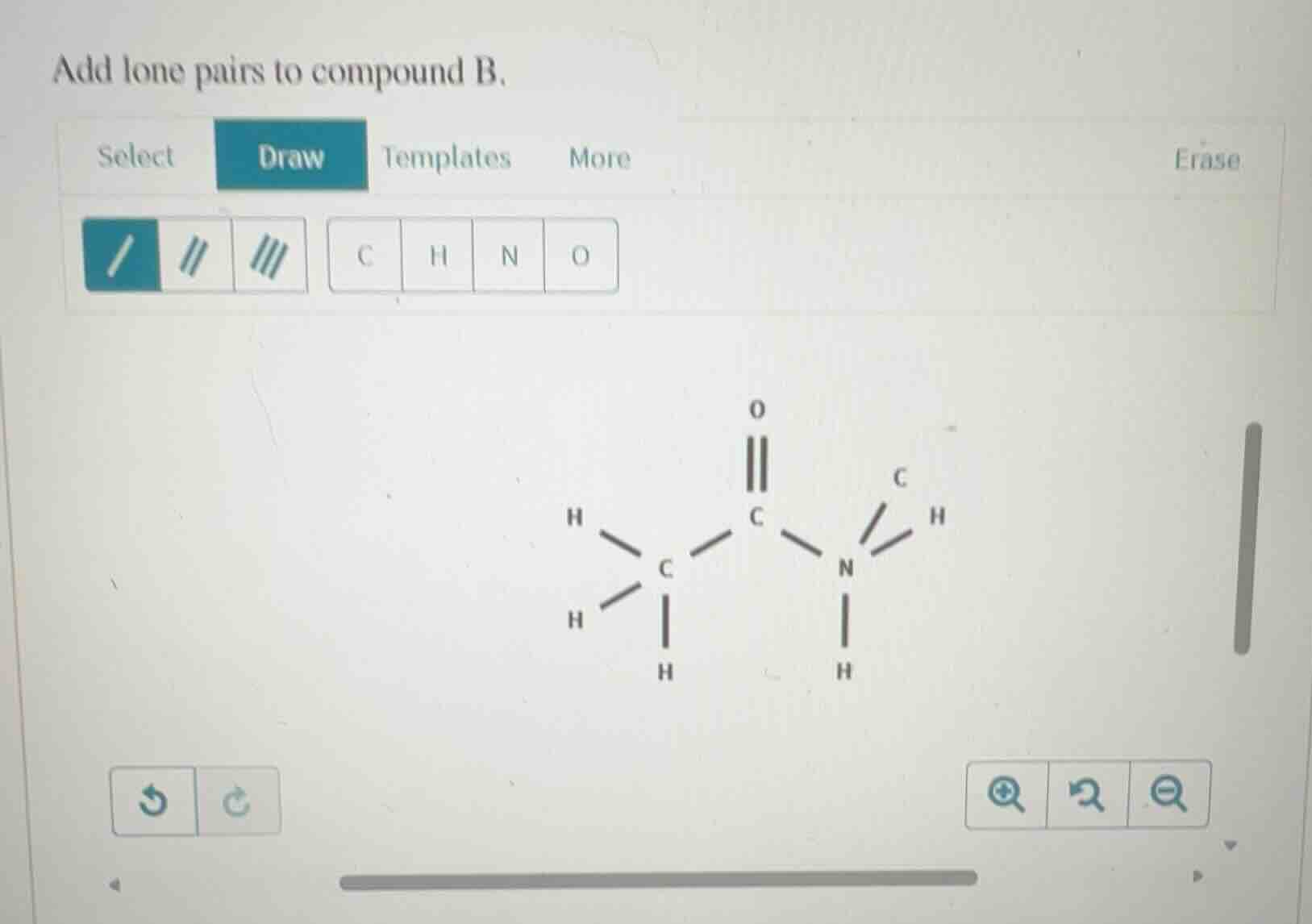
Question
add lone pairs to compound b.
Step1: Analyze oxygen's valence electrons
Oxygen has 6 valence electrons. It forms a double bond (2 electrons shared) with carbon, so remaining electrons: $6 - 2 = 4$, which is 2 lone pairs.
Step2: Analyze nitrogen's valence electrons
Nitrogen has 5 valence electrons. It forms 3 single bonds (3 electrons shared) with 2 H and 1 C, so remaining electrons: $5 - 3 = 2$, which is 1 lone pair.
Step3: Add lone pairs to atoms
Add 2 lone pairs to the oxygen atom, and 1 lone pair to the nitrogen atom.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The structure with lone pairs added has:
- 2 pairs of non-bonding electrons (lone pairs) on the double-bonded oxygen atom
- 1 pair of non-bonding electrons (lone pair) on the nitrogen atom
(Visual representation: The O will have two sets of two dots, and the N will have one set of two dots attached to them, while the existing bonds remain unchanged.)