QUESTION IMAGE
Question
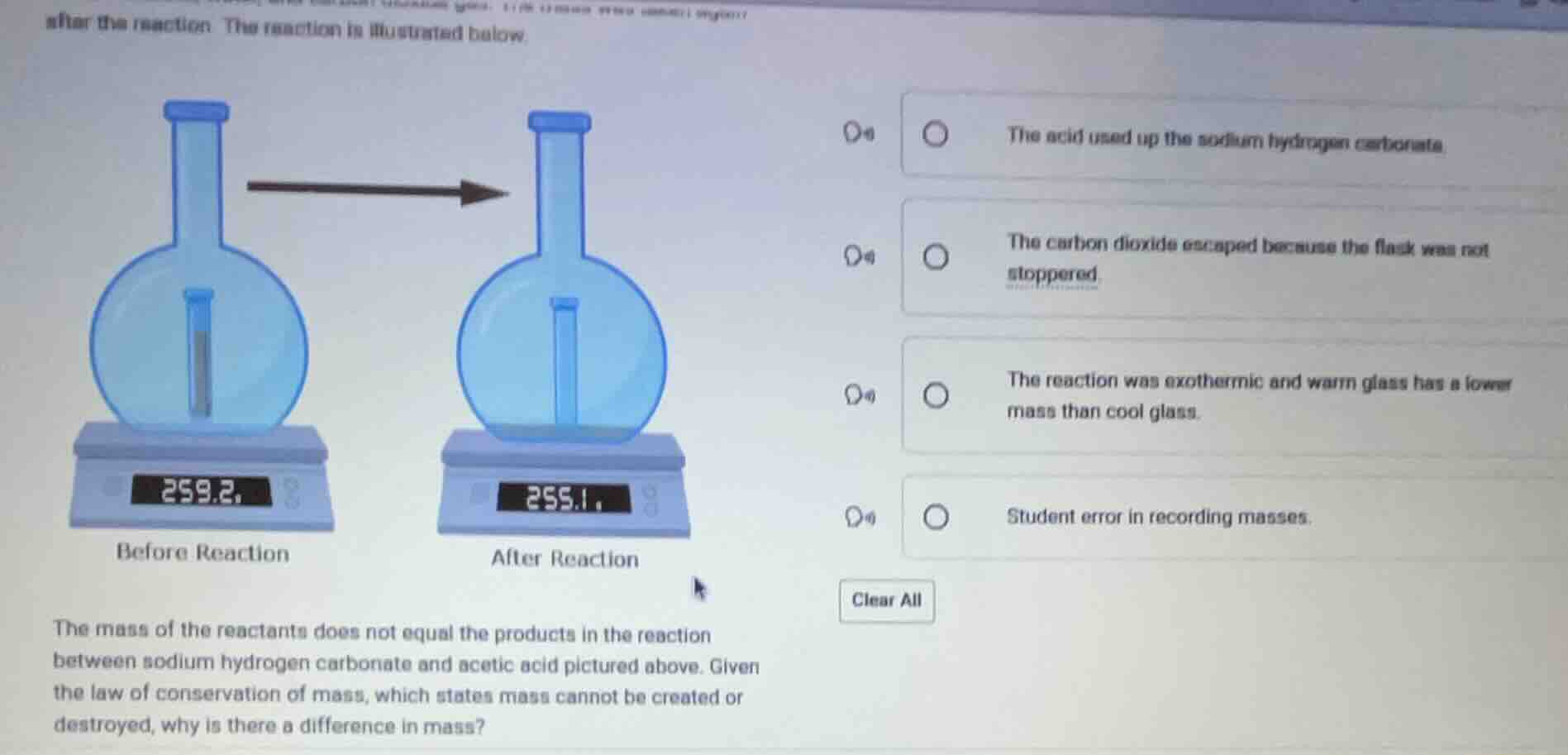
after the reaction. the reaction is illustrated below.
the mass of the reactants does not equal the products in the reaction between sodium hydrogen carbonate and acetic acid pictured above. given the law of conservation of mass, which states mass cannot be created or destroyed, why is there a difference in mass?
options:
- the acid used up the sodium hydrogen carbonate
- the carbon dioxide escaped because the flask was not stoppered
- the reaction was exothermic and warm glass has a lower mass than cool glass
- student error in recording masses
The reaction between sodium hydrogen carbonate and acetic acid produces carbon dioxide gas ($\text{CO}_2$). The law of conservation of mass holds, but the open flask allows this gaseous product to escape the system being weighed, leading to a measured decrease in mass. The other options are incorrect: reactant consumption does not reduce total mass in a closed system, glass mass does not change with temperature in this way, and student error is not supported by the experimental setup shown.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. The carbon dioxide escaped because the flask was not stoppered.