QUESTION IMAGE
Question
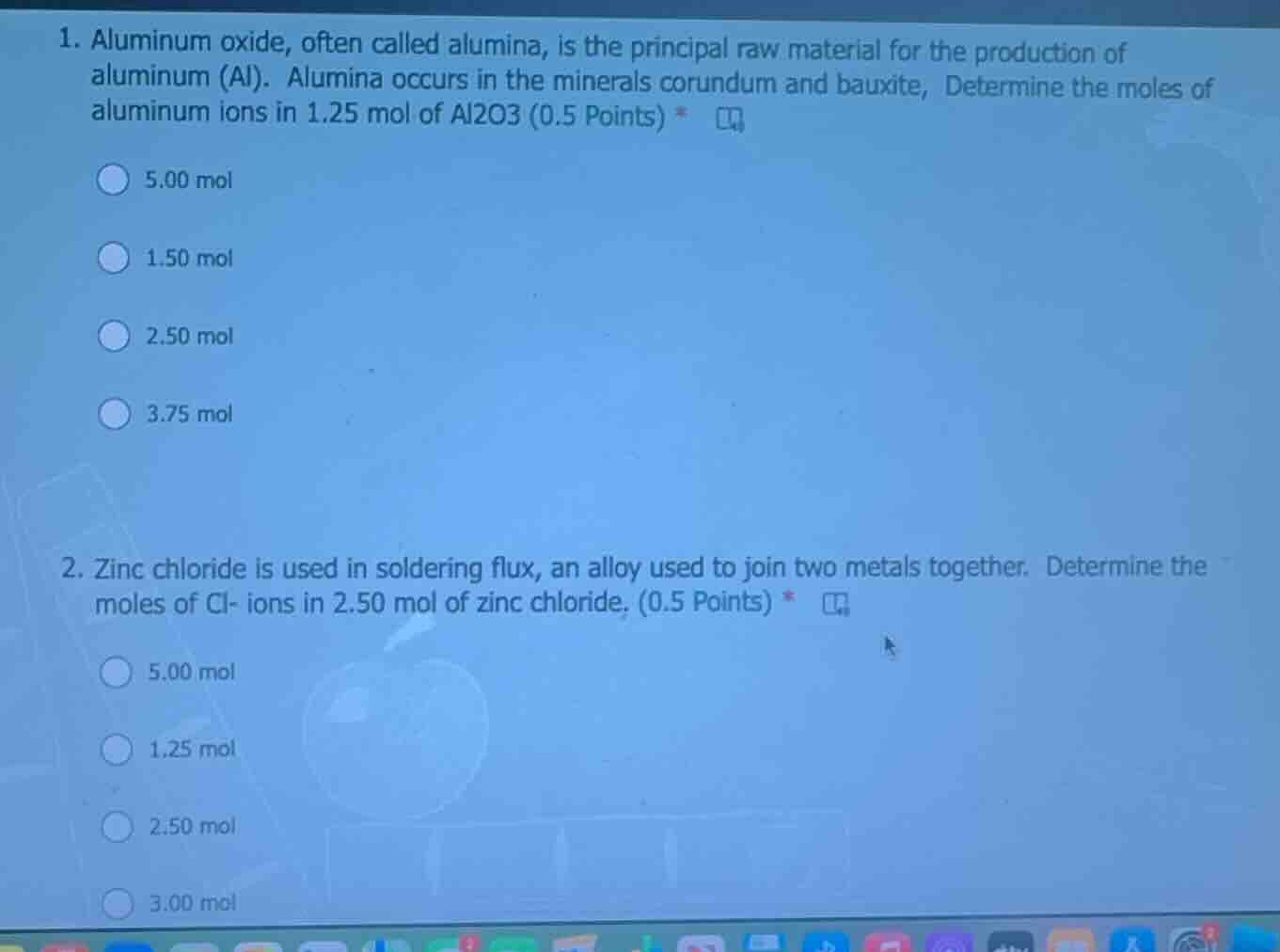
- aluminum oxide, often called alumina, is the principal raw material for the production of aluminum (al). alumina occurs in the minerals corundum and bauxite, determine the moles of aluminum ions in 1.25 mol of al2o3 (0.5 points) *
5.00 mol
1.50 mol
2.50 mol
3.75 mol
- zinc chloride is used in soldering flux, an alloy used to join two metals together. determine the moles of cl- ions in 2.50 mol of zinc chloride. (0.5 points) *
5.00 mol
1.25 mol
2.50 mol
3.00 mol
Question 1
Step1: Analyze the formula of \( \text{Al}_2\text{O}_3 \)
In one mole of \( \text{Al}_2\text{O}_3 \), there are 2 moles of \( \text{Al}^{3+} \) ions (from the subscript 2 on Al in the formula).
Step2: Calculate moles of \( \text{Al}^{3+} \)
Given moles of \( \text{Al}_2\text{O}_3 = 1.25 \, \text{mol} \).
Moles of \( \text{Al}^{3+} = 2 \times \) moles of \( \text{Al}_2\text{O}_3 \)
\( = 2 \times 1.25 \, \text{mol} = 2.50 \, \text{mol} \)
Step1: Analyze the formula of zinc chloride (\( \text{ZnCl}_2 \))
In one mole of \( \text{ZnCl}_2 \), there are 2 moles of \( \text{Cl}^- \) ions (from the subscript 2 on Cl in the formula).
Step2: Calculate moles of \( \text{Cl}^- \)
Given moles of \( \text{ZnCl}_2 = 2.50 \, \text{mol} \).
Moles of \( \text{Cl}^- = 2 \times \) moles of \( \text{ZnCl}_2 \)
\( = 2 \times 2.50 \, \text{mol} = 5.00 \, \text{mol} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2.50 mol (corresponding to the option "2.50 mol")