QUESTION IMAGE
Question
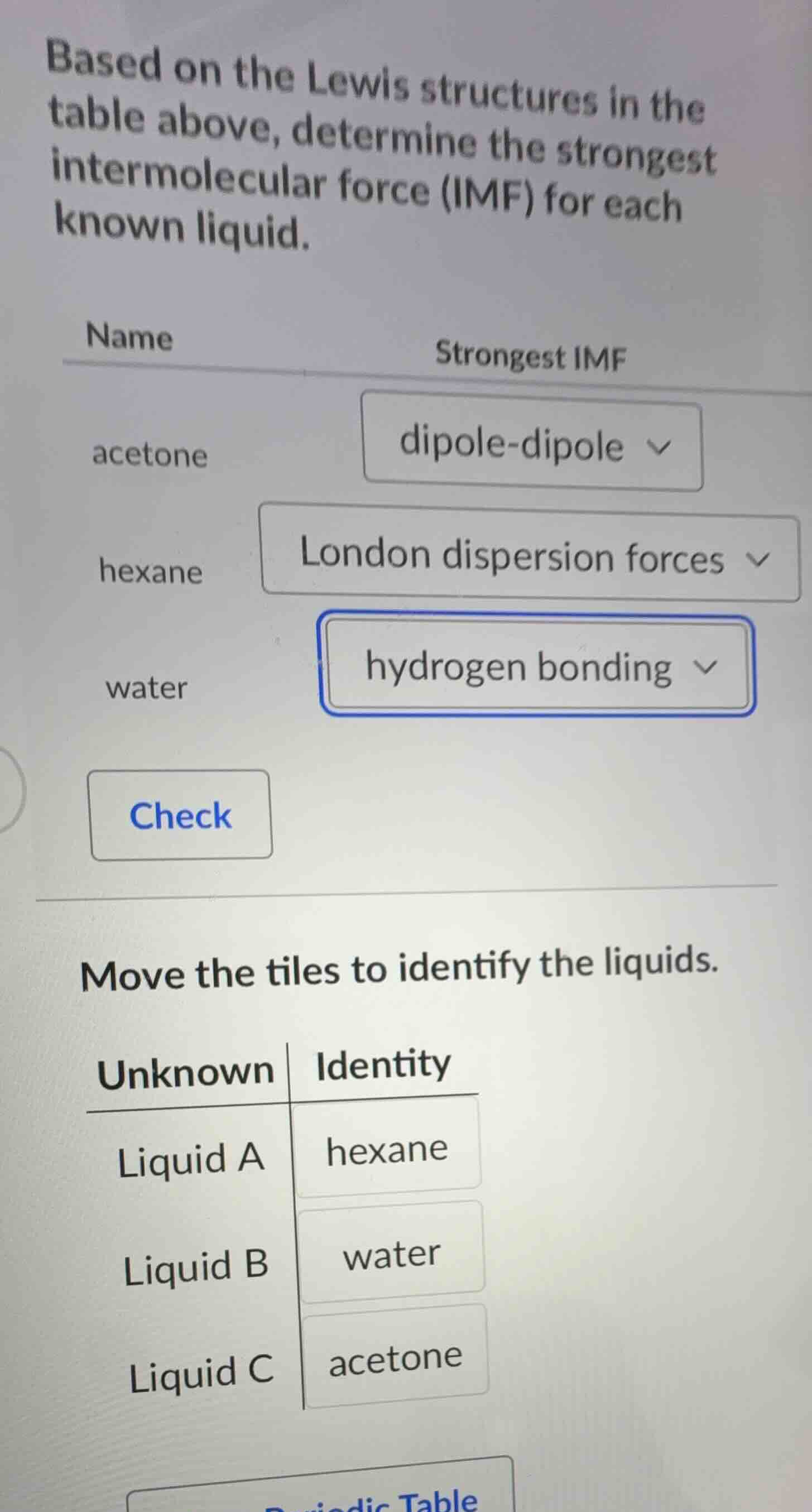
based on the lewis structures in the table above, determine the strongest intermolecular force (imf) for each known liquid.
name\tstrongest imf
acetone\tdipole - dipole
hexane\tlondon dispersion forces
water\thydrogen bonding
check
move the tiles to identify the liquids.
unknown\tidentity
liquid a\thexane
liquid b\twater
liquid c\tacetone
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
acetone: dipole-dipole
hexane: London dispersion forces
water: hydrogen bonding
Liquid A: hexane
Liquid B: water
Liquid C: acetone