QUESTION IMAGE
Question
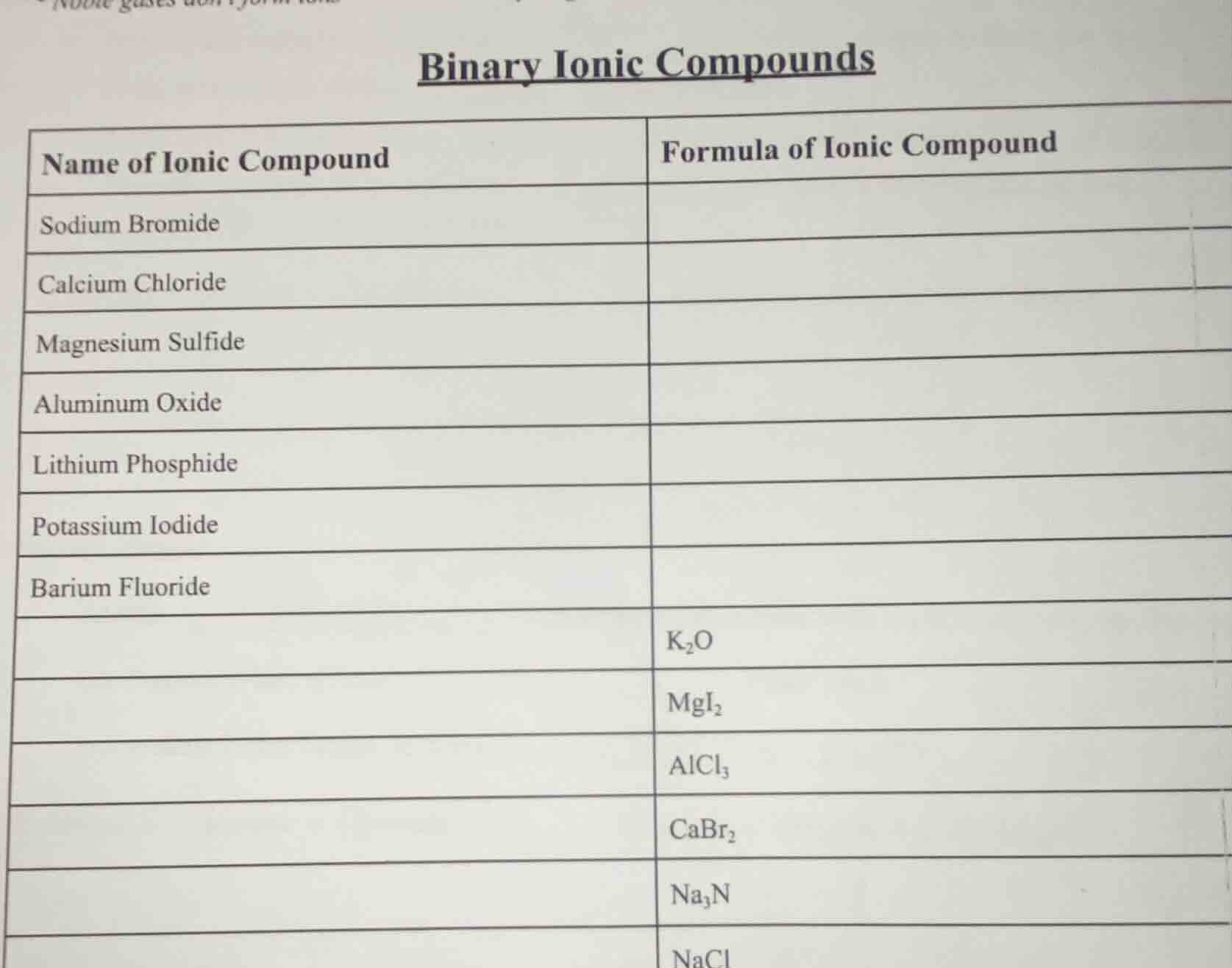
binary ionic compounds
name of ionic compound | formula of ionic compound
sodium bromide |
calcium chloride |
magnesium sulfide |
aluminum oxide |
lithium phosphide |
potassium iodide |
barium fluoride |
| $\ce{k_{2}o}$
| $\ce{mgi_{2}}$
| $\ce{alcl_{3}}$
| $\ce{cabr_{2}}$
| $\ce{na_{3}n}$
| $\ce{nacl}$
To solve for the formulas of binary ionic compounds (and vice versa), we use the principle of charge balance: the total positive charge from cations equals the total negative charge from anions.
1. Sodium Bromide
- Sodium ion: $\text{Na}^+$ (charge = +1)
- Bromide ion: $\text{Br}^-$ (charge = -1)
- Charge balance: $1(+1) + 1(-1) = 0$ → Formula: $\text{NaBr}$
2. Calcium Chloride
- Calcium ion: $\text{Ca}^{2+}$ (charge = +2)
- Chloride ion: $\text{Cl}^-$ (charge = -1)
- Charge balance: $1(+2) + 2(-1) = 0$ → Formula: $\text{CaCl}_2$
3. Magnesium Sulfide
- Magnesium ion: $\text{Mg}^{2+}$ (charge = +2)
- Sulfide ion: $\text{S}^{2-}$ (charge = -2)
- Charge balance: $1(+2) + 1(-2) = 0$ → Formula: $\text{MgS}$
4. Aluminum Oxide
- Aluminum ion: $\text{Al}^{3+}$ (charge = +3)
- Oxide ion: $\text{O}^{2-}$ (charge = -2)
- Charge balance: $2(+3) + 3(-2) = 0$ → Formula: $\text{Al}_2\text{O}_3$
5. Lithium Phosphide
- Lithium ion: $\text{Li}^+$ (charge = +1)
- Phosphide ion: $\text{P}^{3-}$ (charge = -3)
- Charge balance: $3(+1) + 1(-3) = 0$ → Formula: $\text{Li}_3\text{P}$
6. Potassium Iodide
- Potassium ion: $\text{K}^+$ (charge = +1)
- Iodide ion: $\text{I}^-$ (charge = -1)
- Charge balance: $1(+1) + 1(-1) = 0$ → Formula: $\text{KI}$
7. Barium Fluoride
- Barium ion: $\text{Ba}^{2+}$ (charge = +2)
- Fluoride ion: $\text{F}^-$ (charge = -1)
- Charge balance: $1(+2) + 2(-1) = 0$ → Formula: $\text{BaF}_2$
8. Name for $\boldsymbol{\text{K}_2\text{O}}$
- Cation: $\text{K}^+$ (potassium ion)
- Anion: $\text{O}^{2-}$ (oxide ion)
- Name: Potassium Oxide
9. Name for $\boldsymbol{\text{MgI}_2}$
- Cation: $\text{Mg}^{2+}$ (magnesium ion)
- Anion: $\text{I}^-$ (iodide ion)
- Name: Magnesium Iodide
10. Name for $\boldsymbol{\text{AlCl}_3}$
- Cation: $\text{Al}^{3+}$ (aluminum ion)
- Anion: $\text{Cl}^-$ (chloride ion)
- Name: Aluminum Chloride
11. Name for $\boldsymbol{\text{CaBr}_2}$
- Cation: $\text{Ca}^{2+}$ (calcium ion)
- Anion: $\text{Br}^-$ (bromide ion)
- Name: Calcium Bromide
12. Name for $\boldsymbol{\text{Na}_3\text{N}}$
- Cation: $\text{Na}^+$ (sodium ion)
- Anion: $\text{N}^{3-}$ (nitride ion)
- Name: Sodium Nitride
13. Name for $\boldsymbol{\text{NaCl}}$
- Cation: $\text{Na}^+$ (sodium ion)
- Anion: $\text{Cl}^-$ (chloride ion)
- Name: Sodium Chloride
Final Table (Filled)
| Name of Ionic Compound | Formula of Ionic Compound |
|---|---|
| Calcium Chloride | $\text{CaCl}_2$ |
| Magnesium Sulfide | $\text{MgS}$ |
| Aluminum Oxide | $\text{Al}_2\text{O}_3$ |
| Lithium Phosphide | $\text{Li}_3\text{P}$ |
| Potassium Iodide | $\text{KI}$ |
| Barium Fluoride | $\text{BaF}_2$ |
| Potassium Oxide | $\text{K}_2\text{O}$ |
| Magnesium Iodide | $\text{MgI}_2$ |
| Aluminum Chloride | $\text{AlCl}_3$ |
| Calcium Bromide | $\text{CaBr}_2$ |
| Sodium Nitride | $\text{Na}_3\text{N}$ |
| Sodium Chloride | $\text{NaCl}$ |
(Note: The last row in the original table, “NaCl,” is Sodium Chloride, as filled above.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve for the formulas of binary ionic compounds (and vice versa), we use the principle of charge balance: the total positive charge from cations equals the total negative charge from anions.
1. Sodium Bromide
- Sodium ion: $\text{Na}^+$ (charge = +1)
- Bromide ion: $\text{Br}^-$ (charge = -1)
- Charge balance: $1(+1) + 1(-1) = 0$ → Formula: $\text{NaBr}$
2. Calcium Chloride
- Calcium ion: $\text{Ca}^{2+}$ (charge = +2)
- Chloride ion: $\text{Cl}^-$ (charge = -1)
- Charge balance: $1(+2) + 2(-1) = 0$ → Formula: $\text{CaCl}_2$
3. Magnesium Sulfide
- Magnesium ion: $\text{Mg}^{2+}$ (charge = +2)
- Sulfide ion: $\text{S}^{2-}$ (charge = -2)
- Charge balance: $1(+2) + 1(-2) = 0$ → Formula: $\text{MgS}$
4. Aluminum Oxide
- Aluminum ion: $\text{Al}^{3+}$ (charge = +3)
- Oxide ion: $\text{O}^{2-}$ (charge = -2)
- Charge balance: $2(+3) + 3(-2) = 0$ → Formula: $\text{Al}_2\text{O}_3$
5. Lithium Phosphide
- Lithium ion: $\text{Li}^+$ (charge = +1)
- Phosphide ion: $\text{P}^{3-}$ (charge = -3)
- Charge balance: $3(+1) + 1(-3) = 0$ → Formula: $\text{Li}_3\text{P}$
6. Potassium Iodide
- Potassium ion: $\text{K}^+$ (charge = +1)
- Iodide ion: $\text{I}^-$ (charge = -1)
- Charge balance: $1(+1) + 1(-1) = 0$ → Formula: $\text{KI}$
7. Barium Fluoride
- Barium ion: $\text{Ba}^{2+}$ (charge = +2)
- Fluoride ion: $\text{F}^-$ (charge = -1)
- Charge balance: $1(+2) + 2(-1) = 0$ → Formula: $\text{BaF}_2$
8. Name for $\boldsymbol{\text{K}_2\text{O}}$
- Cation: $\text{K}^+$ (potassium ion)
- Anion: $\text{O}^{2-}$ (oxide ion)
- Name: Potassium Oxide
9. Name for $\boldsymbol{\text{MgI}_2}$
- Cation: $\text{Mg}^{2+}$ (magnesium ion)
- Anion: $\text{I}^-$ (iodide ion)
- Name: Magnesium Iodide
10. Name for $\boldsymbol{\text{AlCl}_3}$
- Cation: $\text{Al}^{3+}$ (aluminum ion)
- Anion: $\text{Cl}^-$ (chloride ion)
- Name: Aluminum Chloride
11. Name for $\boldsymbol{\text{CaBr}_2}$
- Cation: $\text{Ca}^{2+}$ (calcium ion)
- Anion: $\text{Br}^-$ (bromide ion)
- Name: Calcium Bromide
12. Name for $\boldsymbol{\text{Na}_3\text{N}}$
- Cation: $\text{Na}^+$ (sodium ion)
- Anion: $\text{N}^{3-}$ (nitride ion)
- Name: Sodium Nitride
13. Name for $\boldsymbol{\text{NaCl}}$
- Cation: $\text{Na}^+$ (sodium ion)
- Anion: $\text{Cl}^-$ (chloride ion)
- Name: Sodium Chloride
Final Table (Filled)
| Name of Ionic Compound | Formula of Ionic Compound |
|---|---|
| Calcium Chloride | $\text{CaCl}_2$ |
| Magnesium Sulfide | $\text{MgS}$ |
| Aluminum Oxide | $\text{Al}_2\text{O}_3$ |
| Lithium Phosphide | $\text{Li}_3\text{P}$ |
| Potassium Iodide | $\text{KI}$ |
| Barium Fluoride | $\text{BaF}_2$ |
| Potassium Oxide | $\text{K}_2\text{O}$ |
| Magnesium Iodide | $\text{MgI}_2$ |
| Aluminum Chloride | $\text{AlCl}_3$ |
| Calcium Bromide | $\text{CaBr}_2$ |
| Sodium Nitride | $\text{Na}_3\text{N}$ |
| Sodium Chloride | $\text{NaCl}$ |
(Note: The last row in the original table, “NaCl,” is Sodium Chloride, as filled above.)