QUESTION IMAGE
Question
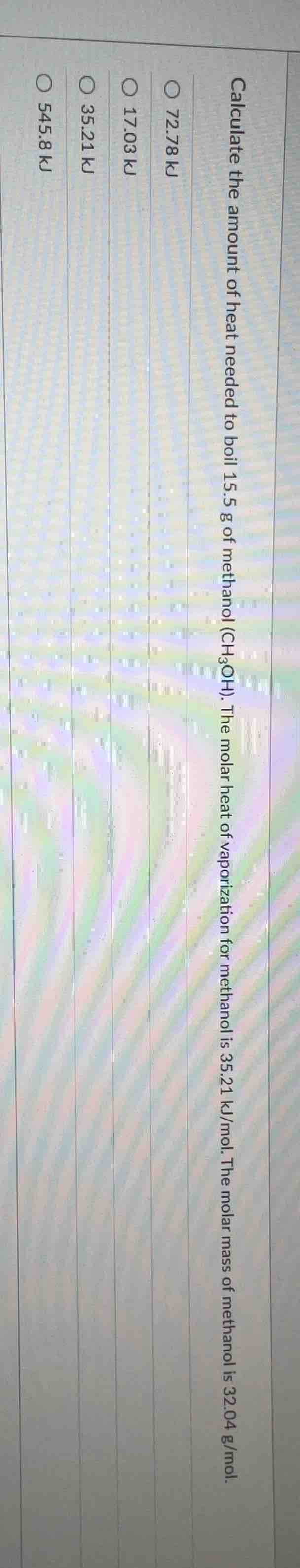
calculate the amount of heat needed to boil 15.5 g of methanol (ch₃oh). the molar heat of vaporization for methanol is 35.21 kj/mol. the molar mass of methanol is 32.04 g/mol.
○ 72.78 kj
○ 17.03 kj
○ 35.21 kj
○ 545.8 kj
Step1: Calculate moles of methanol
Moles = $\frac{\text{mass}}{\text{molar mass}}$, mass = 15.5 g, molar mass = 32.04 g/mol.
Moles = $\frac{15.5}{32.04} \approx 0.4837$ mol.
Step2: Calculate heat required
Heat ($q$) = moles × molar heat of vaporization.
Molar heat of vaporization = 35.21 kJ/mol.
$q = 0.4837 \times 35.21 \approx 17.03$ kJ.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
17.03 kJ (Option: 17.03 kJ)