QUESTION IMAGE
Question
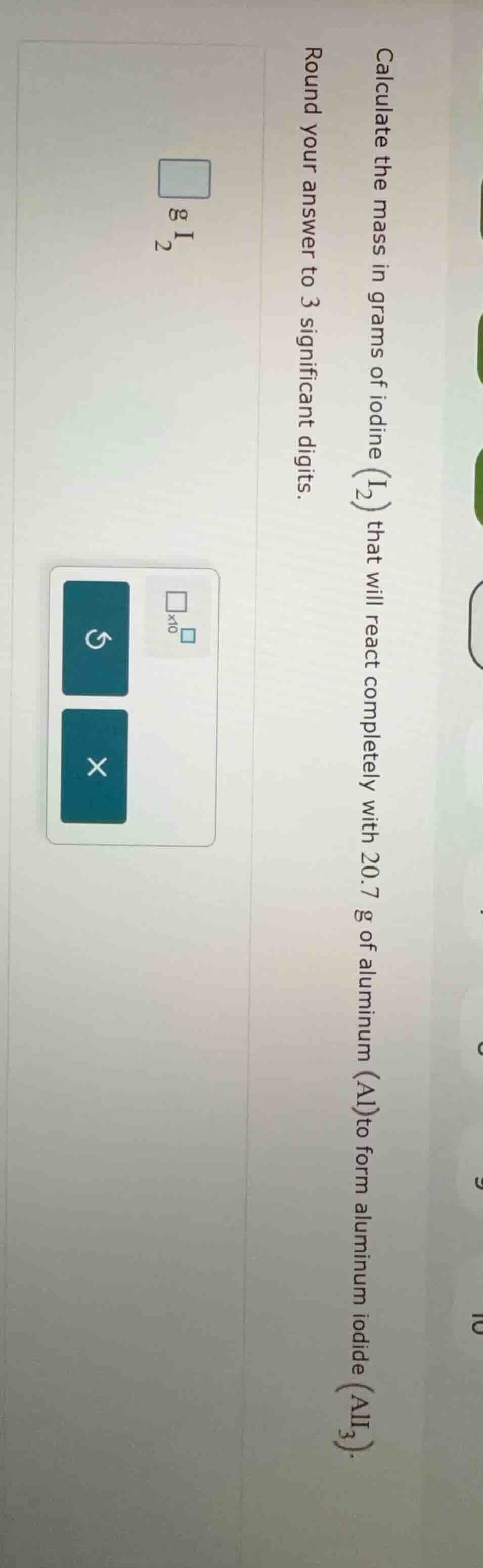
calculate the mass in grams of iodine (i₂) that will react completely with 20.7 g of aluminum (al) to form aluminum iodide (ali₃). round your answer to 3 significant digits.
Step1: Write the balanced chemical equation
The reaction between aluminum (Al) and iodine ($\ce{I2}$) to form aluminum iodide ($\ce{AlI3}$) is:
Step2: Calculate moles of Al
Molar mass of Al is $26.98\ \text{g/mol}$.
Moles of Al = $\frac{\text{mass of Al}}{\text{molar mass of Al}} = \frac{20.7\ \text{g}}{26.98\ \text{g/mol}} \approx 0.767\ \text{mol}$
Step3: Determine moles of $\ce{I2}$ from stoichiometry
From the balanced equation, the mole ratio of $\ce{Al : I2}$ is $2:3$.
So, moles of $\ce{I2} = \frac{3}{2} \times$ moles of Al
Moles of $\ce{I2} = \frac{3}{2} \times 0.767\ \text{mol} \approx 1.1505\ \text{mol}$
Step4: Calculate mass of $\ce{I2}$
Molar mass of $\ce{I2}$ is $2 \times 126.90\ \text{g/mol} = 253.80\ \text{g/mol}$.
Mass of $\ce{I2} = \text{moles of } \ce{I2} \times \text{molar mass of } \ce{I2}$
Mass of $\ce{I2} = 1.1505\ \text{mol} \times 253.80\ \text{g/mol} \approx 292\ \text{g}$ (rounded to 3 significant digits)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
292