QUESTION IMAGE
Question
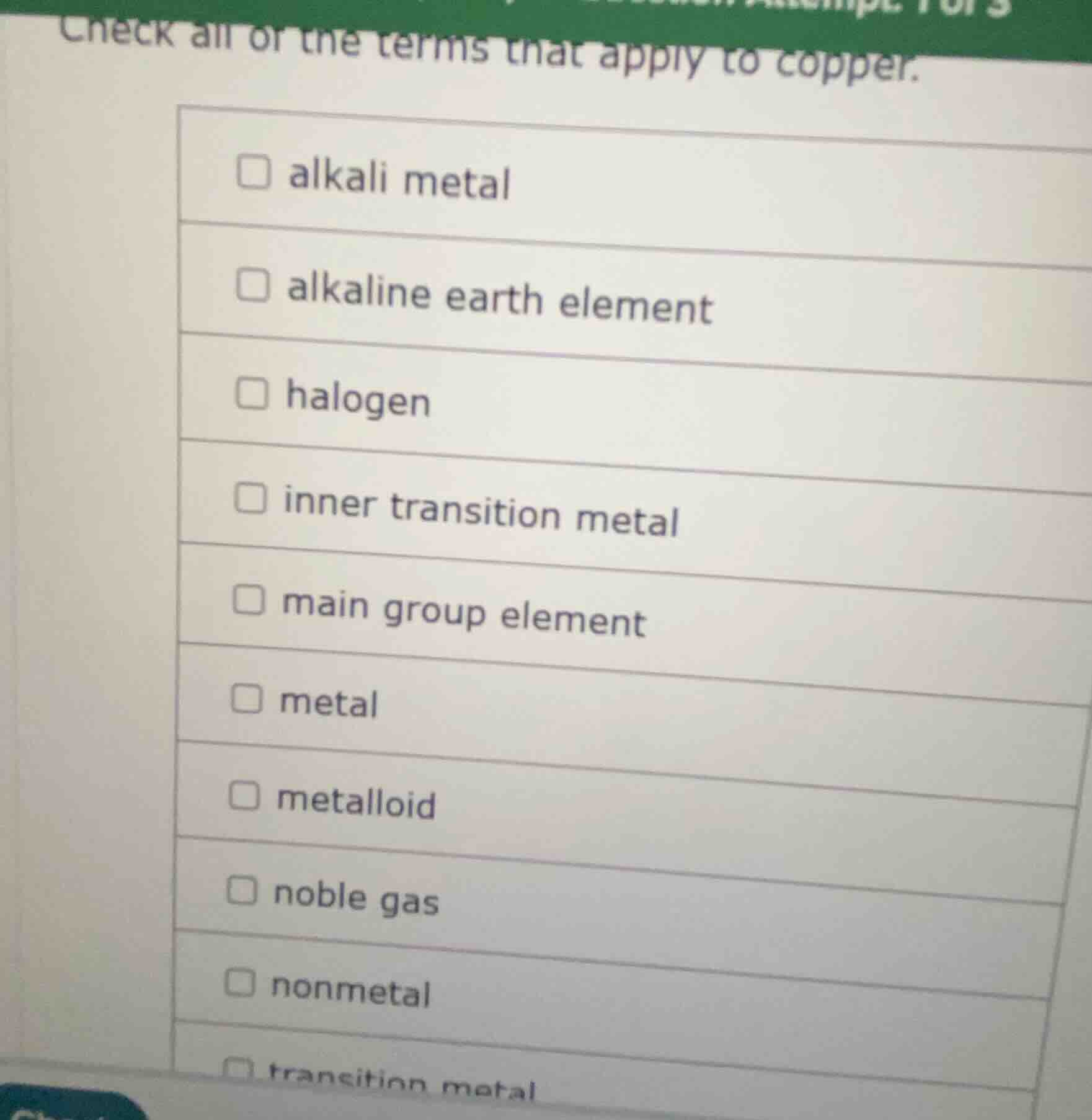
check all of the terms that apply to copper.
☐ alkali metal
☐ alkaline earth element
☐ halogen
☐ inner transition metal
☐ main group element
☐ metal
☐ metalloid
☐ noble gas
☐ nonmetal
☐ transition metal
Brief Explanations
- Alkali metal: Group 1 elements (Li, Na, K, etc.). Copper (Cu) is in Group 11, so no.
- Alkaline earth element: Group 2 elements (Be, Mg, Ca, etc.). Copper is in Group 11, so no.
- Halogen: Group 17 elements (F, Cl, Br, etc.). Copper is a metal, not a halogen, so no.
- Inner transition metal: Lanthanides or actinides (f - block). Copper is in the d - block, so no.
- Main group element: Groups 1, 2, 13 - 18. Copper is in Group 11 (transition metal), so no.
- Metal: Copper has metallic properties (conducts heat/electricity, malleable, ductile), so yes.
- Metalloid: Elements with properties between metal and non - metal (B, Si, Ge, etc.). Copper is a metal, not a metalloid, so no.
- Noble gas: Group 18 elements (He, Ne, Ar, etc.). Copper is a metal, so no.
- Nonmetal: Elements lacking metallic properties. Copper is a metal, so no.
- Transition metal: d - block elements (Groups 3 - 12). Copper is in Group 11 (d - block), so yes (note: the last option in the visible part is "transition metal", so we check that too as copper is a transition metal).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
metal, transition metal