QUESTION IMAGE
Question
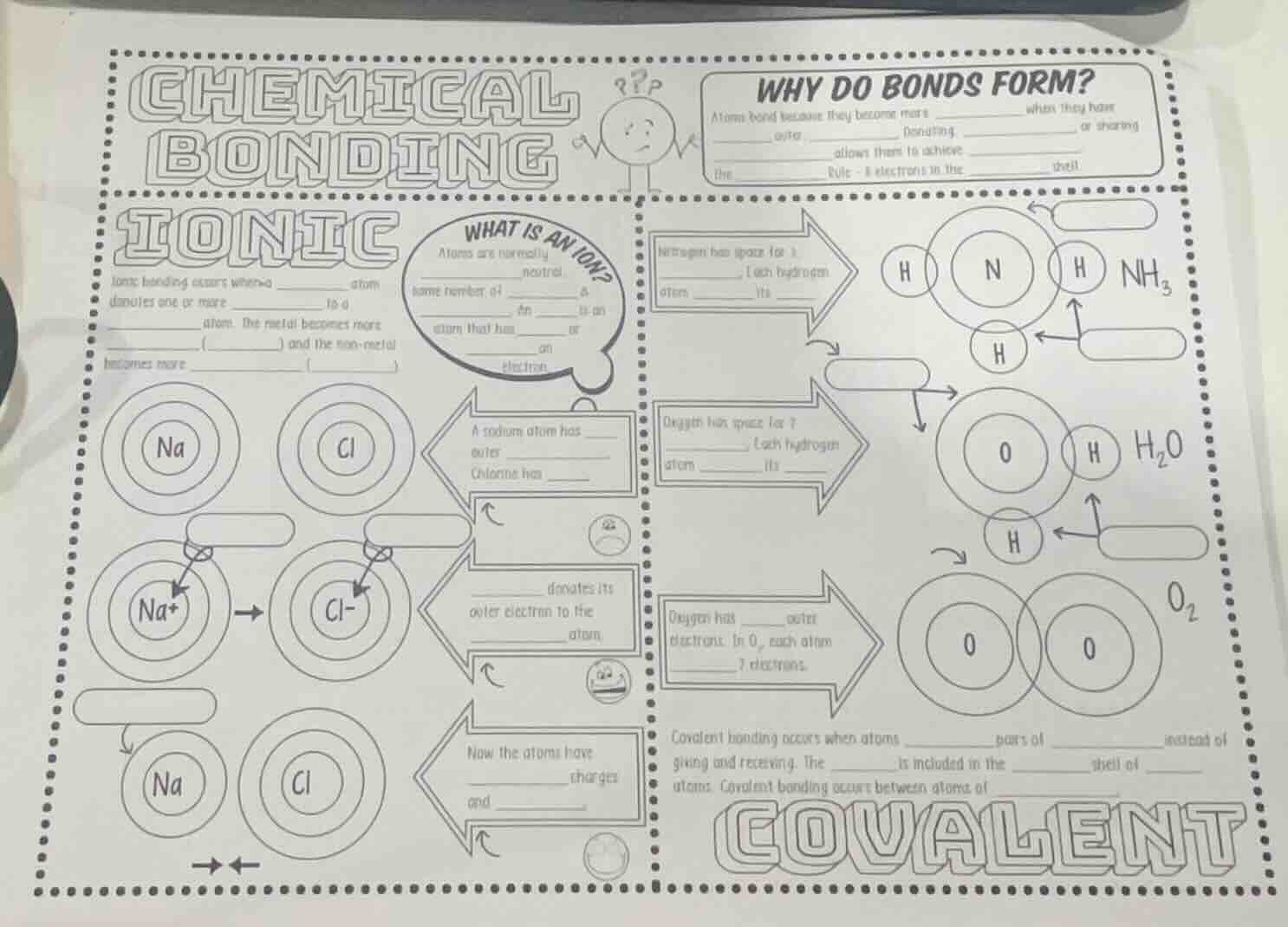
chemical bonding
why do bonds form?
atoms bond because they become more ____ when they have __ after __ (bonding, __ or sharing __ allows them to achieve __ (the __ rule - 8 electrons in the ____ shell)
ionic
ionic bonding occurs when a ____ atom donates one or more __ to a __ atom. the metal becomes more __ (__) and the non - metal becomes more __ (____)
what is an ion?
atoms are normally ____ (neutral) (same number of __ & __). an __ is an atom that has __ or ____ electrons
a sodium atom has ____ outer __ chlorine has ____
____ donates its outer electron to the ____ atom
now the atoms have ____ charges (__ and ____)
nitrogen has space for 3 ____ (each hydrogen atom __ its ____)
oxygen has space for 2 ____ (each hydrogen atom __ its ____)
oxygen has ____ outer electrons. in o₂, each atom ____ electrons
covalent bonding occurs when atoms ____ pairs of __ instead of giving and receiving. the __ is included in the __ shell of __ atoms. covalent bonding occurs between atoms of ____
To solve the fill - in - the - blanks related to chemical bonding, we analyze each part based on chemical bonding concepts:
Part 1: Why do bonds form?
Atoms bond because they become more stable when they have completed their outer shell by losing, gaining or sharing electrons. This allows them to achieve the octet rule - 8 electrons in the outer shell (or the duet rule for hydrogen, 2 electrons in the first shell).
Part 2: Ionic Bonding
Ionic bonding occurs when a metal atom donates one or more electrons to a non - metal atom. The metal becomes more positive (a cation) and the non - metal becomes more negative (an anion).
What is an ion?
Atoms are normally electrically neutral (same number of protons & electrons). An ion is an atom that has gained or lost an electron. A sodium atom has 1 outer electron. Chlorine has 7 outer electrons. Sodium donates its outer electron to the chlorine atom. Now the atoms have opposite charges (positive and negative).
Part 3: Covalent Bonding
Ammonia ($NH_3$)
Nitrogen has space for 3 electrons (each hydrogen atom shares its electron).
Water ($H_2O$)
Oxygen has space for 2 electrons (each hydrogen atom shares its electron).
Oxygen ($O_2$)
Oxygen has 6 outer electrons. In $O_2$, each atom shares 2 electrons.
Covalent bonding occurs when atoms share pairs of electrons instead of giving and receiving. The shared electron is included in the outer shell of both atoms. Covalent bonding occurs between atoms of non - metals.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s (filling the blanks):
Why do bonds form?
- stable
- completed
- outer
- losing
- gaining
- electrons
- octet
- outer
Ionic Bonding
- metal
- electrons
- non - metal
- positive
- cation
- negative
- anion
What is an ion?
- electrically
- protons
- electrons
- gained
- lost
- 1
- electron
- 7
- outer electrons
- Sodium
- chlorine
- opposite
- positive
- negative
Covalent Bonding (Ammonia)
- electrons
- shares
- electron
Covalent Bonding (Water)
- electrons
- shares
- electron
Covalent Bonding (Oxygen)
- 6
- shares
- 2
General Covalent Bonding
- share
- electrons
- shared electron
- outer
- both
- non - metals