QUESTION IMAGE
Question
the chemical formula for methanol is $ce{ch_{3}oh}$. which of the following models could be used to represent methanol?
a
image of a molecular model
b
image of a molecular model
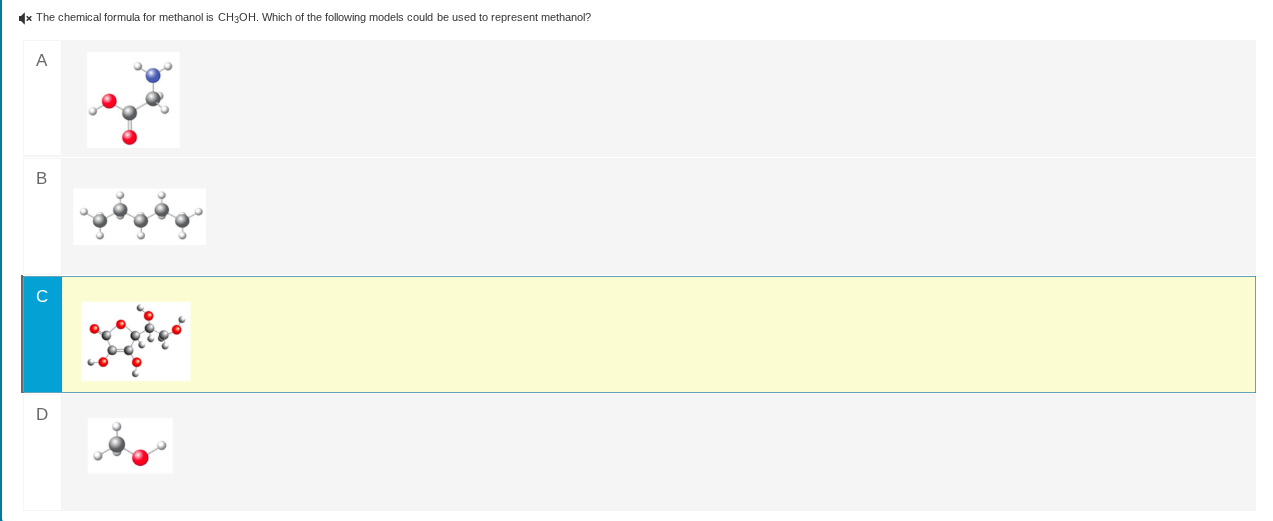
c
image of a molecular model
d
image of a molecular model
Brief Explanations
Methanol has the formula \( \text{CH}_3\text{OH} \), which consists of 1 carbon (C), 4 hydrogens (H), and 1 oxygen (O). Let's analyze each option:
- Option A: Contains a blue atom (not part of methanol's composition) and multiple red/gray atoms, not matching \( \text{CH}_3\text{OH} \).
- Option B: Shows a chain of carbon atoms (like an alkane, e.g., ethane or longer), no oxygen, so incorrect.
- Option C: Depicts a larger molecular structure (likely a polymer or carbohydrate - like), not a single methanol molecule.
- Option D: Has 1 gray (C), 3 white (H) attached to C, 1 red (O) with 1 white (H) attached to O – matching \( \text{CH}_3\text{OH} \)’s atom count and structure.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. The model with 1 carbon (gray), 3 hydrogens (white) on carbon, 1 oxygen (red) with 1 hydrogen (white) on oxygen.