QUESTION IMAGE
Question
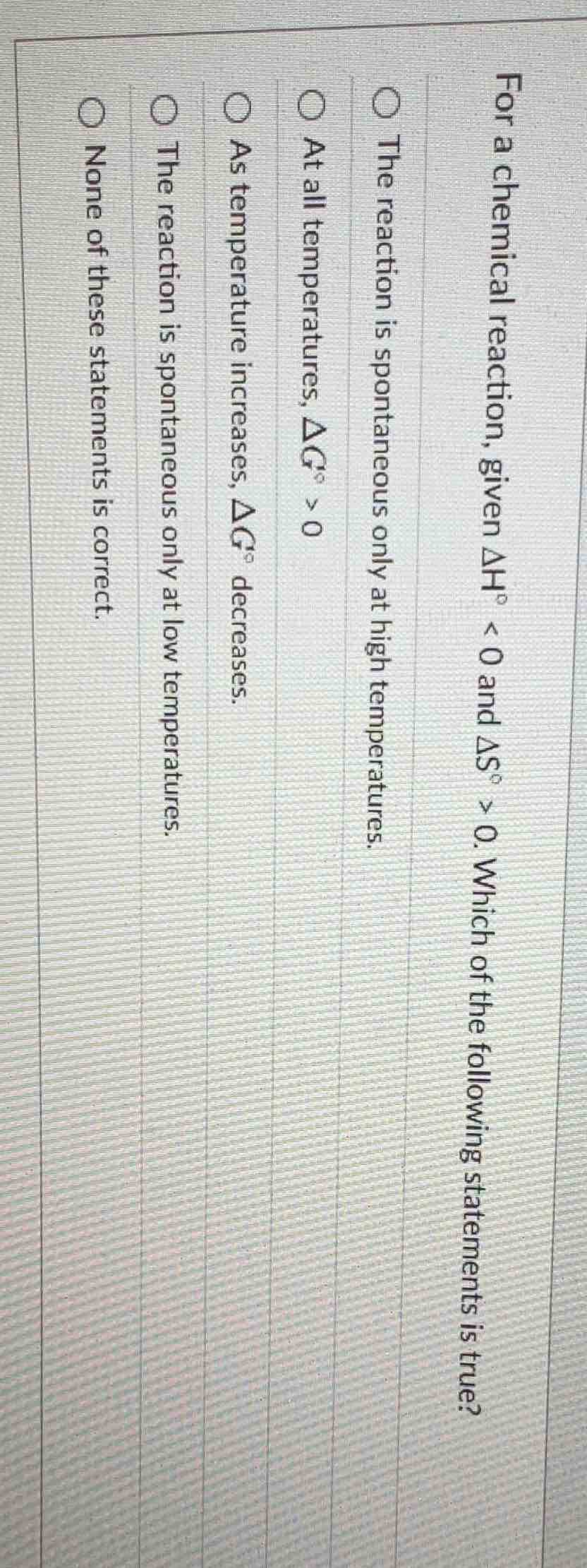
for a chemical reaction, given $delta h^{circ} < 0$ and $delta s^{circ} > 0$. which of the following statements is true?
the reaction is spontaneous only at high temperatures.
at all temperatures, $delta g^{circ} > 0$
as temperature increases, $delta g^{circ}$ decreases.
the reaction is spontaneous only at low temperatures.
none of these statements is correct.
To determine the correct statement, we use the Gibbs free energy equation: $\Delta G^\circ = \Delta H^\circ - T\Delta S^\circ$. Given $\Delta H^\circ < 0$ (exothermic) and $\Delta S^\circ > 0$ (increase in disorder).
- For the first option: A reaction is spontaneous when $\Delta G^\circ < 0$. With $\Delta H^\circ < 0$ and $\Delta S^\circ > 0$, $-T\Delta S^\circ$ is negative (since $T$ is positive and $\Delta S^\circ > 0$), so $\Delta G^\circ = (\text{negative}) - T(\text{positive}) = \text{more negative}$. Thus, the reaction is spontaneous at all temperatures, not just high, so this is wrong.
- Second option: $\Delta G^\circ = \Delta H^\circ - T\Delta S^\circ$. Since $\Delta H^\circ < 0$ and $\Delta S^\circ > 0$, $-T\Delta S^\circ$ is negative. So $\Delta G^\circ$ will be negative (because a negative minus a positive - but actually, $\Delta H^\circ$ is negative, and $-T\Delta S^\circ$ is negative, so adding two negatives? Wait, no: $\Delta G^\circ = \Delta H^\circ - T\Delta S^\circ$. If $\Delta H^\circ$ is negative and $\Delta S^\circ$ is positive, then $-T\Delta S^\circ$ is negative (because $T$ is positive, $\Delta S^\circ$ positive, so $T\Delta S^\circ$ positive, then $-T\Delta S^\circ$ negative). So $\Delta G^\circ = (\text{negative}) + (\text{negative})$? Wait, no, subtraction: $\Delta G^\circ = \Delta H^\circ + (-T\Delta S^\circ)$. So $\Delta H^\circ$ is negative, $-T\Delta S^\circ$ is negative (since $T\Delta S^\circ$ is positive, so negative of that is negative). Wait, no, that can't be. Wait, $\Delta S^\circ > 0$, so $T\Delta S^\circ$ is positive (since $T > 0$ in Kelvin). Then $-T\Delta S^\circ$ is negative. So $\Delta G^\circ = \Delta H^\circ + (-T\Delta S^\circ) = (\text{negative}) + (\text{negative})$? No, wait, $\Delta H^\circ$ is negative, and we subtract $T\Delta S^\circ$ (which is positive, because $T>0$ and $\Delta S^\circ>0$). So $\Delta G^\circ = (\text{negative}) - (\text{positive})$. But since $\Delta H^\circ$ is negative and $T\Delta S^\circ$ is positive, let's take numbers: suppose $\Delta H^\circ = -10$, $\Delta S^\circ = 5$, $T = 298$. Then $\Delta G^\circ = -10 - 298*5 = -10 - 1490 = -1500 < 0$. So $\Delta G^\circ$ is negative, not positive. So the second option is wrong.
- Third option: As $T$ increases, $-T\Delta S^\circ$ becomes more negative (since $T$ is larger, $T\Delta S^\circ$ is larger, so $-T\Delta S^\circ$ is more negative). $\Delta H^\circ$ is constant (standard enthalpy). So $\Delta G^\circ = \Delta H^\circ - T\Delta S^\circ$. As $T$ increases, $-T\Delta S^\circ$ becomes more negative, so $\Delta G^\circ$ becomes more negative (decreases, since it's getting more negative). So this statement is correct: "As temperature increases, $\Delta G^\circ$ decreases."
- Fourth option: The reaction is spontaneous at all temperatures (because $\Delta G^\circ < 0$ always), not just low, so this is wrong.
- Fifth option: Since the third option is correct, this is wrong.
So the correct statement is "As temperature increases, $\Delta G^\circ$ decreases."
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
As temperature increases, $\Delta G^\circ$ decreases. (The option with this statement)