QUESTION IMAGE
Question
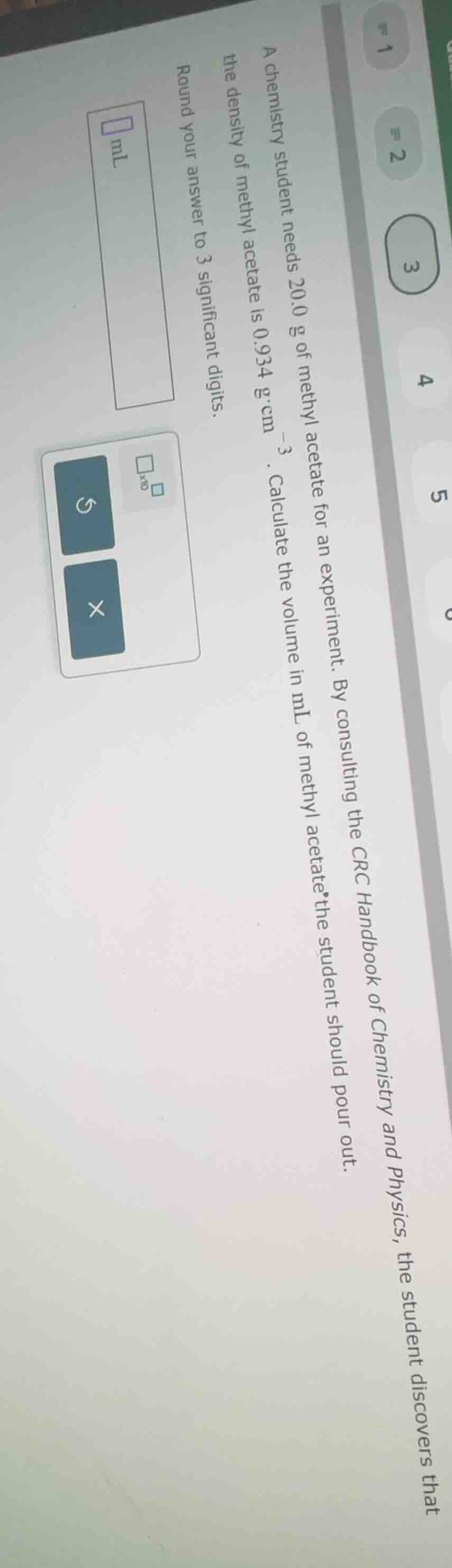
a chemistry student needs 20.0 g of methyl acetate for an experiment. by consulting the crc handbook of chemistry and physics, the student discovers that the density of methyl acetate is 0.934 g·cm⁻³. calculate the volume in ml of methyl acetate the student should pour out. round your answer to 3 significant digits.
Step1: Recall density formula
Density formula is $
ho=\frac{m}{V}$, so we can rearrange it to find volume: $V = \frac{m}{
ho}$. Here, mass $m = 20.0\space g$, density $
ho=0.934\space g\cdot cm^{-3}$. Also, $1\space cm^{3}=1\space mL$.
Step2: Calculate volume
Substitute values into the formula: $V=\frac{20.0\space g}{0.934\space g\cdot cm^{-3}}$. Calculate this division: $\frac{20.0}{0.934}\approx21.4\space cm^{3}$. Since $1\space cm^{3}=1\space mL$, the volume in mL is also approximately $21.4$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
21.4