QUESTION IMAGE
Question
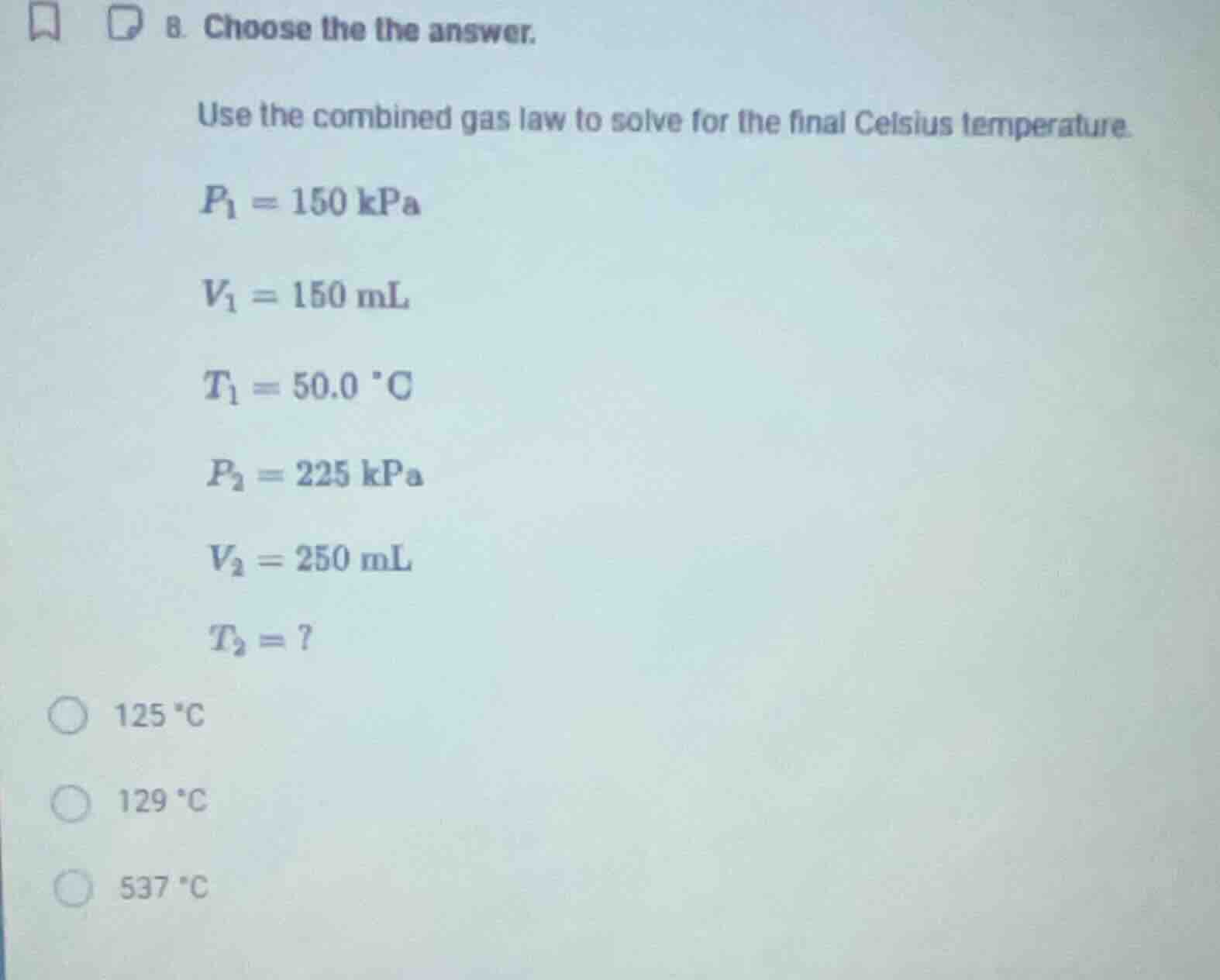
- choose the answer.
use the combined gas law to solve for the final celsius temperature.
$p_1 = 150$ kpa
$v_1 = 150$ ml
$t_1 = 50.0$ °c
$p_2 = 225$ kpa
$v_2 = 250$ ml
$t_2 = ?$
125 °c
129 °c
537 °c
Step1: Convert T₁ to Kelvin
$T_1 = 50.0 + 273.15 = 323.15\ \text{K}$
Step2: Write combined gas law
$$\frac{P_1V_1}{T_1} = \frac{P_2V_2}{T_2}$$
Step3: Rearrange for T₂
$$T_2 = \frac{P_2V_2T_1}{P_1V_1}$$
Step4: Substitute values
$$T_2 = \frac{225\ \text{kPa} \times 250\ \text{mL} \times 323.15\ \text{K}}{150\ \text{kPa} \times 150\ \text{mL}}$$
Step5: Calculate T₂ in Kelvin
$T_2 = 403.9375\ \text{K}$
Step6: Convert T₂ to Celsius
$T_2 = 403.9375 - 273.15 = 130.7875\ ^\circ\text{C} \approx 129\ ^\circ\text{C}$ (rounded to match options)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
129 °C