QUESTION IMAGE
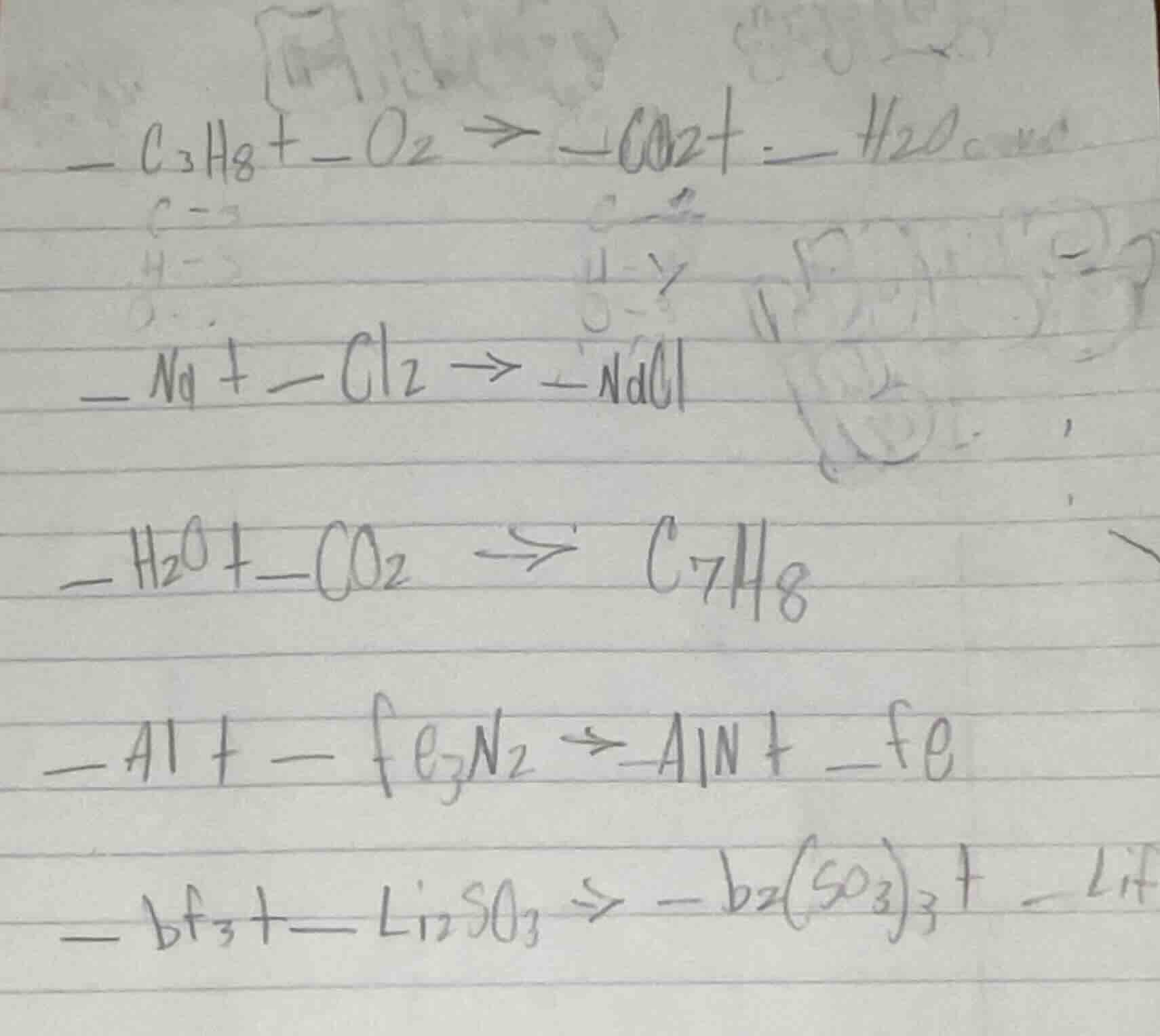
Question
— c₃h₈ + — o₂ → — co₂ + — h₂o
— na + — cl₂ → — nacl
— h₂o + — co₂ → c₇h₈
— al + — fe₃n₂ → — aln + — fe
— bf₃ + — li₂so₃ → — b₂(so₃)₃ + — lif
First Equation: $\boldsymbol{C_3H_8 + O_2
ightarrow CO_2 + H_2O}$
Step1: Balance Carbon (C)
On the left, $C_3H_8$ has 3 C. So, put 3 in front of $CO_2$:
$C_3H_8 + O_2
ightarrow 3CO_2 + H_2O$
Step2: Balance Hydrogen (H)
$C_3H_8$ has 8 H. So, put 4 in front of $H_2O$ (since $4 \times 2 = 8$):
$C_3H_8 + O_2
ightarrow 3CO_2 + 4H_2O$
Step3: Balance Oxygen (O)
Right side: $3CO_2$ has $3 \times 2 = 6$ O; $4H_2O$ has $4 \times 1 = 4$ O. Total O: $6 + 4 = 10$. So, $O_2$ needs $\frac{10}{2} = 5$:
$C_3H_8 + 5O_2
ightarrow 3CO_2 + 4H_2O$
Second Equation: $\boldsymbol{Na + Cl_2
ightarrow NaCl}$
Step1: Balance Chlorine (Cl)
$Cl_2$ has 2 Cl. So, put 2 in front of $NaCl$:
$Na + Cl_2
ightarrow 2NaCl$
Step2: Balance Sodium (Na)
$2NaCl$ has 2 Na. So, put 2 in front of $Na$:
$2Na + Cl_2
ightarrow 2NaCl$
Third Equation: $\boldsymbol{H_2O + CO_2
ightarrow C_7H_8 + O_2}$ (Assuming Product is $C_7H_8 + O_2$, Likely Combustion)
Step1: Balance Carbon (C)
$C_7H_8$ has 7 C. So, put 7 in front of $CO_2$:
$H_2O + 7CO_2
ightarrow C_7H_8 + O_2$
Step2: Balance Hydrogen (H)
$C_7H_8$ has 8 H. So, put 4 in front of $H_2O$ (since $4 \times 2 = 8$):
$4H_2O + 7CO_2
ightarrow C_7H_8 + O_2$
Step3: Balance Oxygen (O)
Left: $4H_2O$ has $4 \times 1 = 4$ O; $7CO_2$ has $7 \times 2 = 14$ O. Total O: $4 + 14 = 18$.
Right: $O_2$ has $2x$ O; $C_7H_8$ has 0 O. So, $2x = 18 \implies x = 9$:
$4H_2O + 7CO_2
ightarrow C_7H_8 + 9O_2$
Fourth Equation: $\boldsymbol{Al + Fe_3N_2
ightarrow AlN + Fe}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Step1: Balance Boron (B)
$B_2(SO_3)_3$ has 2 B. $BF_3$ has 1 B. So, put 2 in front of $BF_3$:
$2BF_3 + Li_2SO_3
ightarrow B_2(SO_3)_3 + LiF$
Step2: Balance Sulfite ($SO_3$)
$B_2(SO_3)_3$ has 3 $SO_3$. So, put 3 in front of $Li_2SO_3$:
$2BF_3 + 3Li_2SO_3
ightarrow B_2(SO_3)_3 + LiF$
Step3: Balance Lithium (Li)
$3Li_2SO_3$ has 6 Li. $LiF$ has 1 Li. So, put 6 in front of $LiF$:
$2BF_3 + 3Li_2SO_3
ightarrow B_2(SO_3)_3 + 6LiF$
Step4: Balance Fluorine (F)
$2BF_3$ has $2 \times 3 = 6$ F. $6LiF$ has 6 F. Balanced.
Final Balanced Equations:
- $\boldsymbol{1C_3H_8 + 5O_2
ightarrow 3CO_2 + 4H_2O}$
- $\boldsymbol{2Na + 1Cl_2
ightarrow 2NaCl}$
- $\boldsymbol{4H_2O + 7CO_2
ightarrow 1C_7H_8 + 9O_2}$ (Adjusted for Combustion)
- $\boldsymbol{2Al + 1Fe_3N_2
ightarrow 2AlN + 3Fe}$
- $\boldsymbol{2BF_3 + 3Li_2SO_3
ightarrow 1B_2(SO_3)_3 + 6LiF}$