QUESTION IMAGE
Question
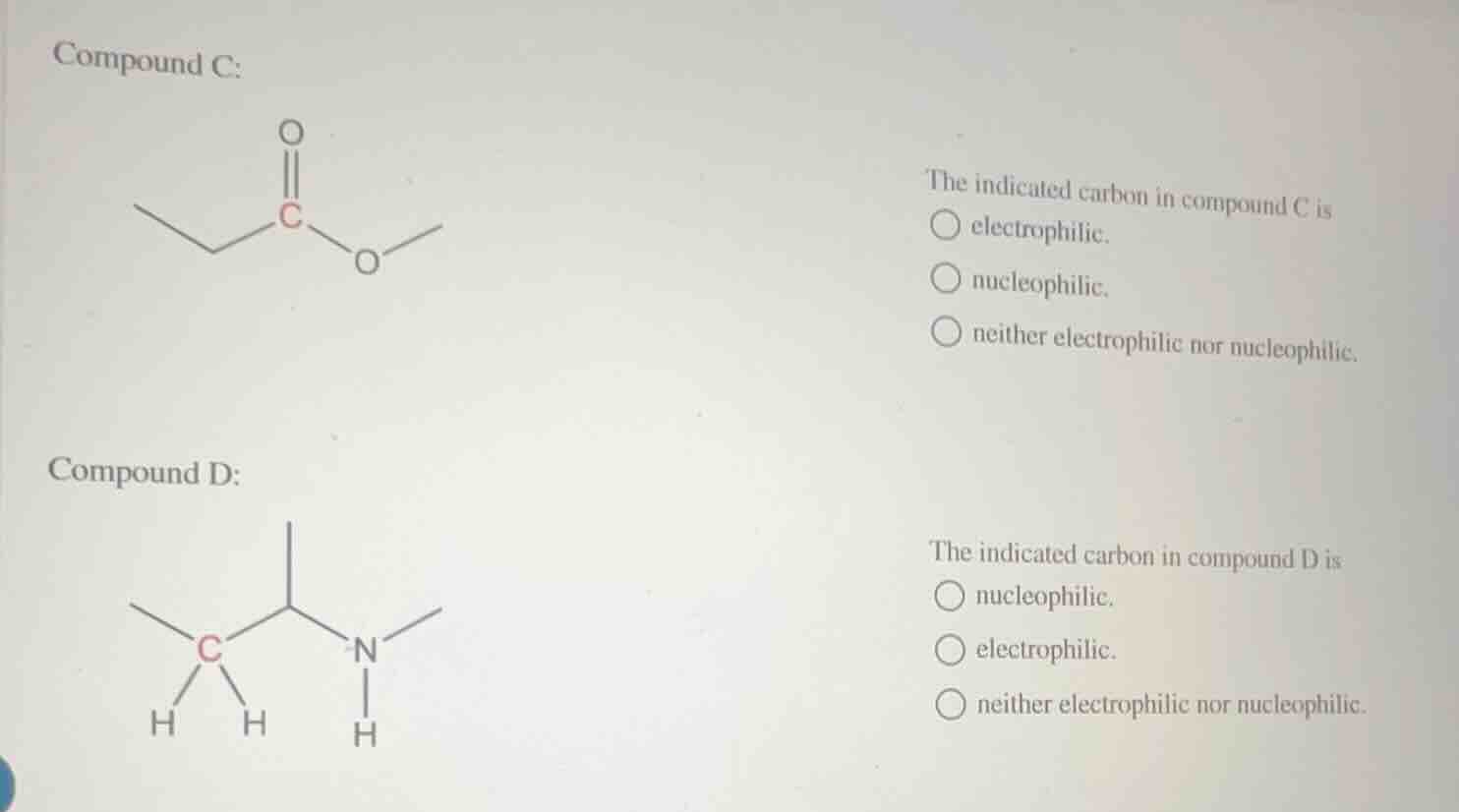
compound c:
the indicated carbon in compound c is
○ electrophilic.
○ nucleophilic.
○ neither electrophilic nor nucleophilic.
compound d:
the indicated carbon in compound d is
○ nucleophilic.
○ electrophilic.
○ neither electrophilic nor nucleophilic.
For Compound C: The indicated carbon is part of a carbonyl group (C=O) bonded to an oxygen atom. Oxygen is highly electronegative, pulling electron density away from the carbonyl carbon, leaving it electron-deficient. Electron-deficient atoms act as electrophiles, attracting electron-rich species.
For Compound D: The indicated carbon is a saturated carbon bonded to two other carbons, two hydrogens, and adjacent to a nitrogen-containing group. Nitrogen is electronegative but does not withdraw enough electron density to make this carbon electron-deficient; the carbon also has no excess electron density to act as a nucleophile. It has a neutral electron distribution.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Compound C: electrophilic.
Compound D: neither electrophilic nor nucleophilic.