QUESTION IMAGE
Question
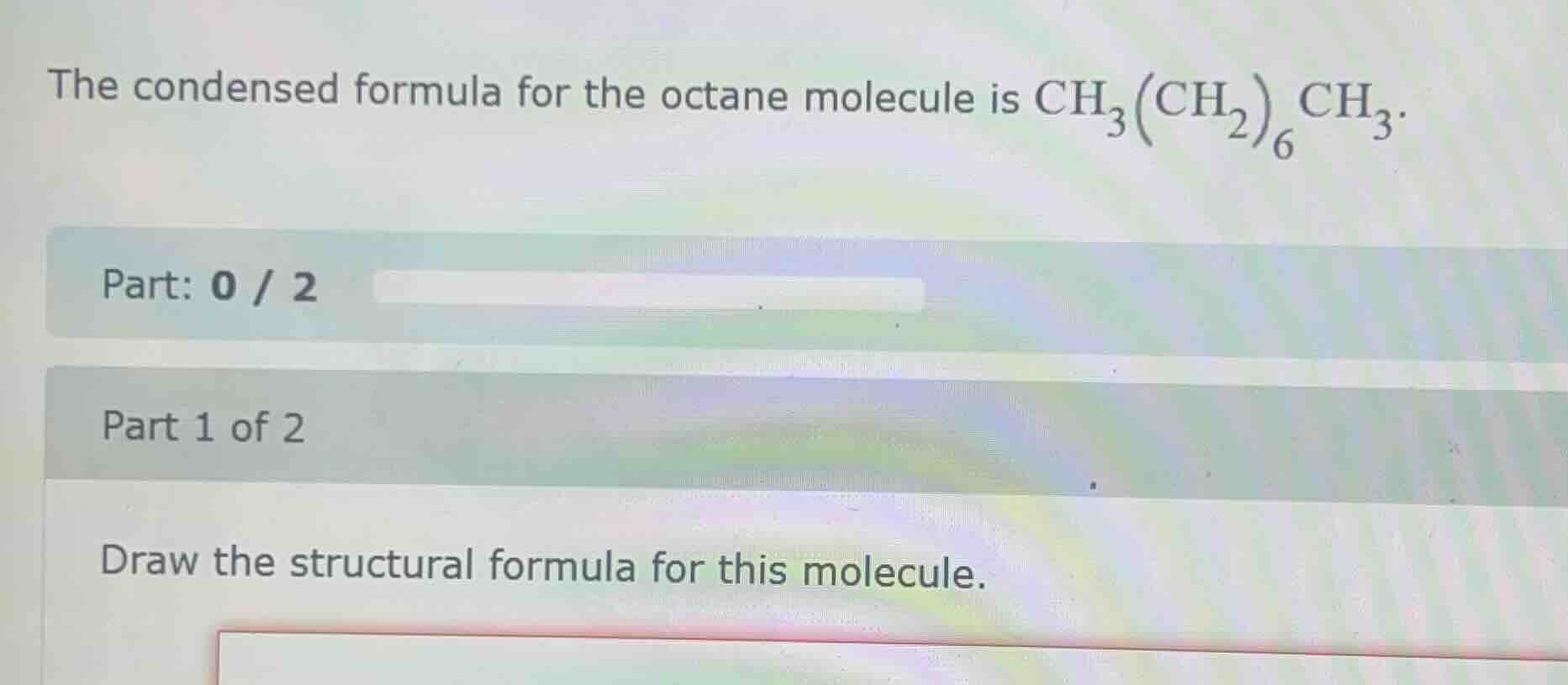
the condensed formula for the octane molecule is $\ce{ch_{3}(ch_{2})_{6}ch_{3}}$.
part: 0 / 2
part 1 of 2
draw the structural formula for this molecule.
Octane is an alkane with 8 carbon atoms in a straight chain. Each terminal carbon is bonded to 3 hydrogen atoms, and each internal carbon is bonded to 2 hydrogen atoms, with single covalent bonds connecting all carbons in a linear chain.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The structural formula (full displayed) is:
$$\text{H}-\underset{\underset{\text{H}}{|}}{\overset{\overset{\text{H}}{|}}{\text{C}}}-\underset{\underset{\text{H}}{|}}{\overset{\overset{\text{H}}{|}}{\text{C}}}-\underset{\underset{\text{H}}{|}}{\overset{\overset{\text{H}}{|}}{\text{C}}}-\underset{\underset{\text{H}}{|}}{\overset{\overset{\text{H}}{|}}{\text{C}}}-\underset{\underset{\text{H}}{|}}{\overset{\overset{\text{H}}{|}}{\text{C}}}-\underset{\underset{\text{H}}{|}}{\overset{\overset{\text{H}}{|}}{\text{C}}}-\underset{\underset{\text{H}}{|}}{\overset{\overset{\text{H}}{|}}{\text{C}}}-\underset{\underset{\text{H}}{|}}{\overset{\overset{\text{H}}{|}}{\text{C}}}-\text{H}$$
Or in simplified line-angle form (common for alkanes):
A straight line with 8 vertices, where each vertex represents a carbon atom (with the corresponding number of hydrogen atoms implied to satisfy carbon's 4-valence requirement).