QUESTION IMAGE
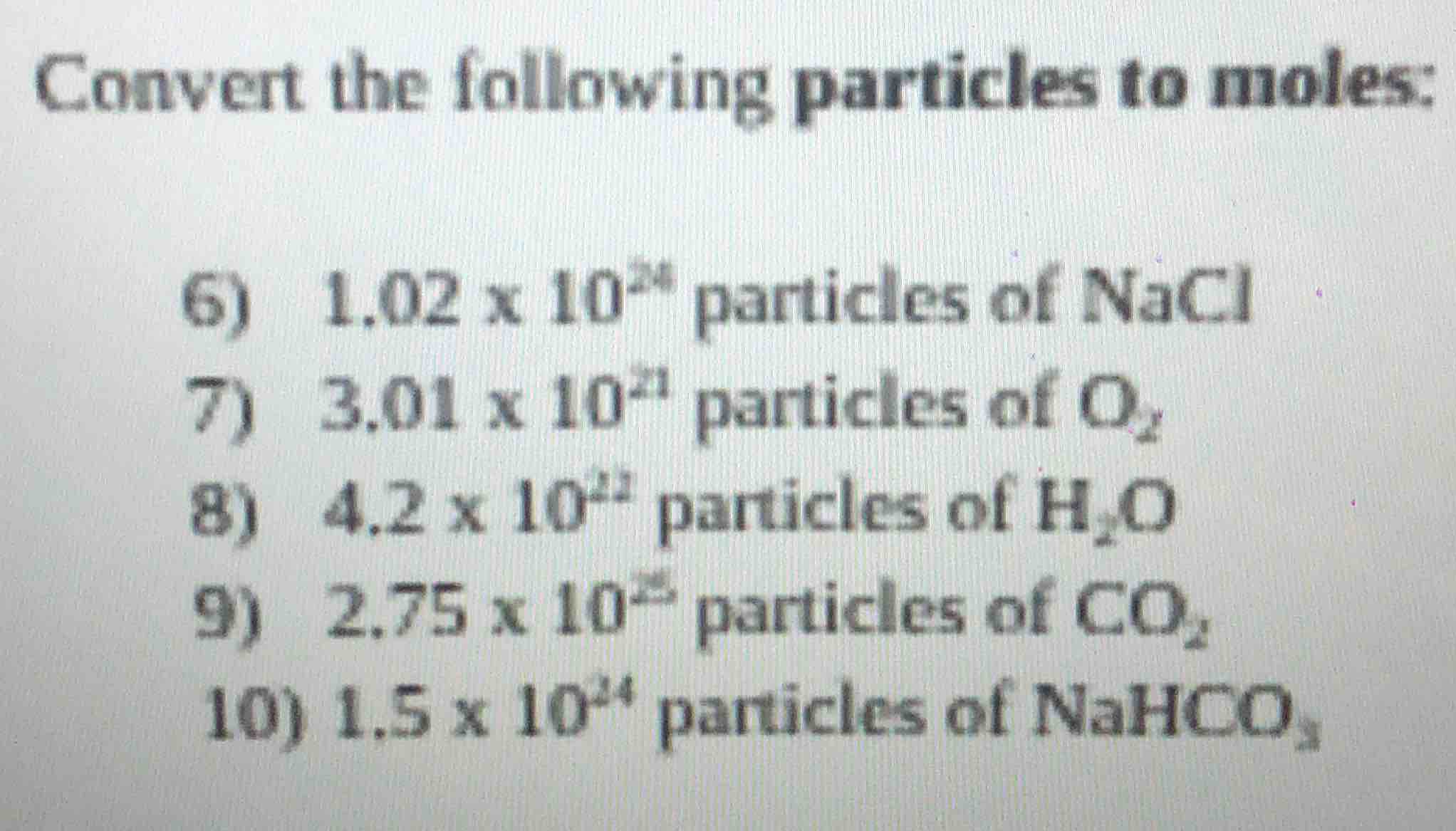
Question
convert the following particles to moles:
- 1.02 x 10²⁴ particles of nacl
- 3.01 x 10²¹ particles of o₂
- 4.2 x 10²² particles of h₂o
- 2.75 x 10²⁵ particles of co₂
- 1.5 x 10²⁴ particles of nahco₃
Problem 6:
Step1: Recall Avogadro's number
Avogadro's number is \( 6.02 \times 10^{23} \) particles per mole. The formula to convert particles to moles is \( \text{Moles} = \frac{\text{Number of particles}}{6.02 \times 10^{23} \text{ particles/mol}} \)
Step2: Substitute the values
For \( 1.02 \times 10^{24} \) particles of \( \text{NaCl} \), we have:
\( \text{Moles} = \frac{1.02 \times 10^{24}}{6.02 \times 10^{23}} \)
Calculate the numerator divided by the denominator: \( \frac{1.02 \times 10^{24}}{6.02 \times 10^{23}} \approx \frac{10.2 \times 10^{23}}{6.02 \times 10^{23}} \approx \frac{10.2}{6.02} \approx 1.69 \) moles
Step1: Use the particle to mole formula
The formula is \( \text{Moles} = \frac{\text{Number of particles}}{6.02 \times 10^{23} \text{ particles/mol}} \)
Step2: Plug in the values
For \( 3.01 \times 10^{21} \) particles of \( \text{O}_2 \):
\( \text{Moles} = \frac{3.01 \times 10^{21}}{6.02 \times 10^{23}} \)
Simplify the exponents and divide the coefficients: \( \frac{3.01}{6.02} \times 10^{21 - 23} = 0.5 \times 10^{-2} = 5 \times 10^{-3} = 0.005 \) moles
Step1: Apply the conversion formula
\( \text{Moles} = \frac{\text{Number of particles}}{6.02 \times 10^{23} \text{ particles/mol}} \)
Step2: Substitute the given particles
For \( 4.2 \times 10^{22} \) particles of \( \text{H}_2\text{O} \):
\( \text{Moles} = \frac{4.2 \times 10^{22}}{6.02 \times 10^{23}} \)
Calculate the division: \( \frac{4.2}{6.02} \times 10^{22 - 23} \approx 0.698 \times 10^{-1} \approx 0.0698 \) moles (or approximately \( 0.07 \) moles)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Approximately \( 1.69 \) moles