QUESTION IMAGE
Question
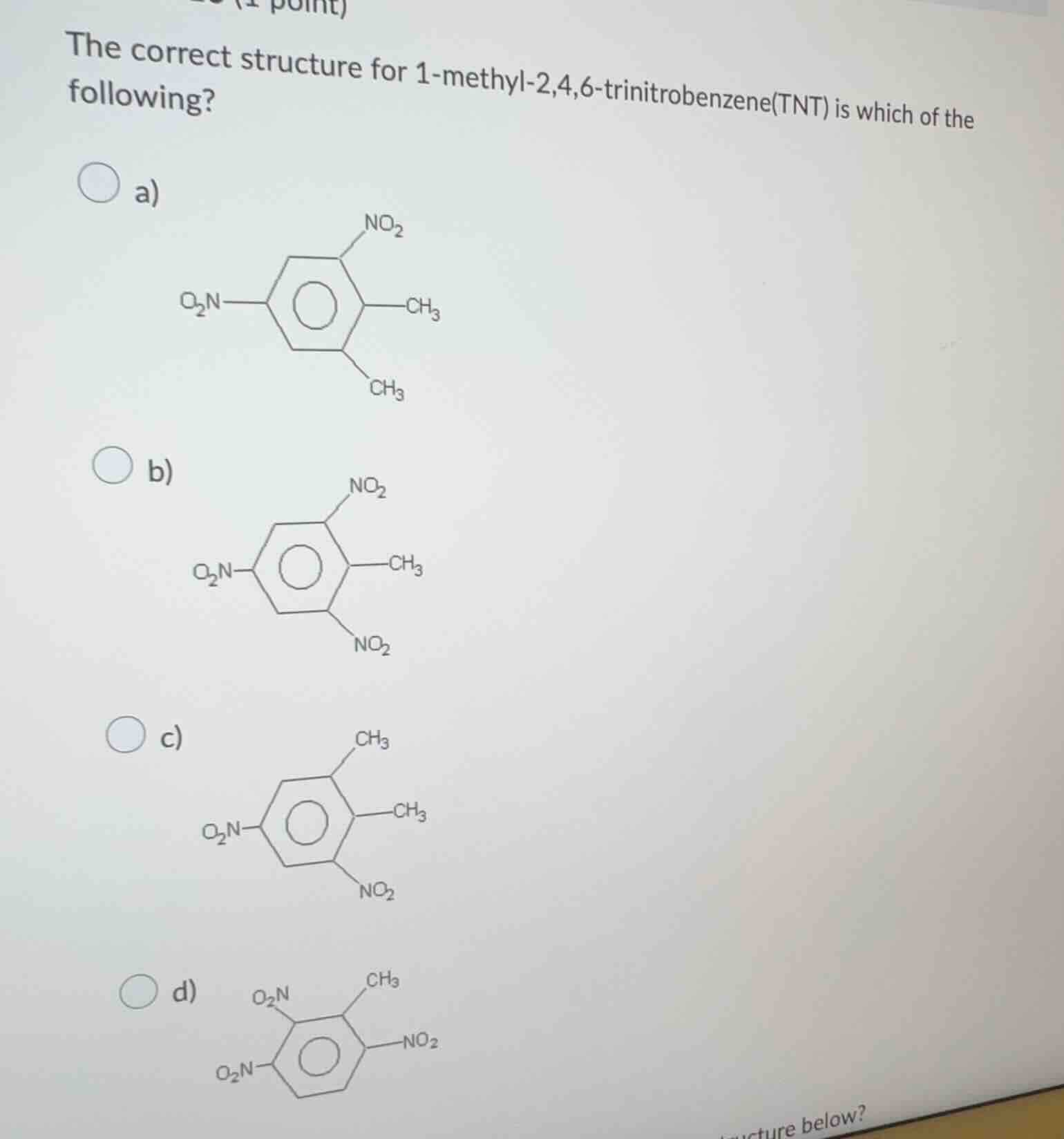
the correct structure for 1 - methyl - 2,4,6 - trinitrobenzene(tnt) is which of the following?
a)
b)
c)
d)
Brief Explanations
To determine the correct structure of 1 - methyl - 2,4,6 - trinitrobenzene (TNT), we analyze the substituent positions on the benzene ring:
- The parent compound is benzene with a methyl group ($\ce{CH_3}$) at position 1.
- Nitro groups ($\ce{NO_2}$) are present at positions 2, 4, and 6.
- Let's analyze each option:
- Option a: Has two methyl groups, which is incorrect as TNT has only one methyl group.
- Option b: The methyl group is at position 1, and nitro groups are at positions 2, 4, and 6 (counting the carbon atoms of the benzene ring, the positions of $\ce{NO_2}$ groups match 2, 4, 6 relative to the $\ce{CH_3}$ group at position 1).
- Option c: The number and positions of nitro groups do not match the formula (it has fewer nitro groups and incorrect positions relative to the methyl groups).
- Option d: The arrangement of nitro groups and the methyl group does not conform to the 2,4,6 - trinitro and 1 - methyl substitution pattern.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) The structure with a $\ce{CH_3}$ group and $\ce{NO_2}$ groups at positions corresponding to 1 - methyl - 2,4,6 - trinitrobenzene.