QUESTION IMAGE
Question
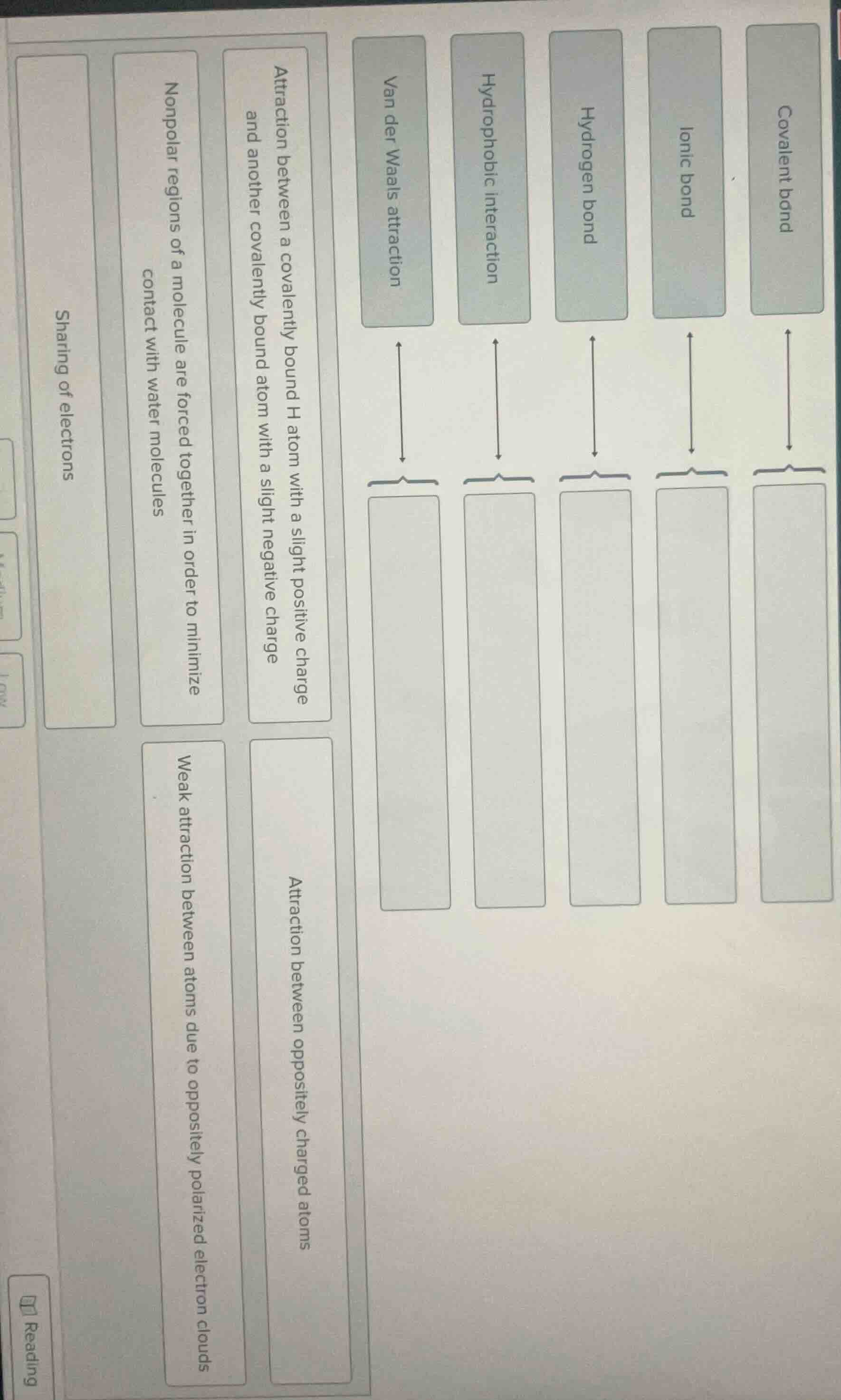
covalent bond
ionic bond
hydrogen bond
hydrophobic interaction
van der waals attraction
attraction between a covalently bound h atom with a slight positive charge and another covalently bound atom with a slight negative charge
nonpolar regions of a molecule are forced together in order to minimize contact with water molecules
sharing of electrons
attraction between oppositely charged atoms
weak attraction between atoms due to oppositely polarized electron clouds
Each bond/interaction is matched to its defining characteristic based on core chemical principles:
- Covalent bonds form when atoms share electrons to fill valence shells.
- Ionic bonds arise from the attraction between atoms with opposite electrical charges.
- Hydrogen bonds occur between a slightly positive covalently bonded hydrogen and a slightly negative covalently bonded atom (like O, N).
- Hydrophobic interactions force nonpolar molecular regions together to reduce contact with polar water molecules.
- Van der Waals attractions are weak, temporary attractions between atoms due to polarized electron clouds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Covalent bond → Sharing of electrons
- Ionic bond → Attraction between oppositely charged atoms
- Hydrogen bond → Attraction between a covalently bound H atom with a slight positive charge and another covalently bound atom with a slight negative charge
- Hydrophobic interaction → Nonpolar regions of a molecule are forced together in order to minimize contact with water molecules
- Van der Waals attraction → Weak attraction between atoms due to oppositely polarized electron clouds