QUESTION IMAGE
Question
date
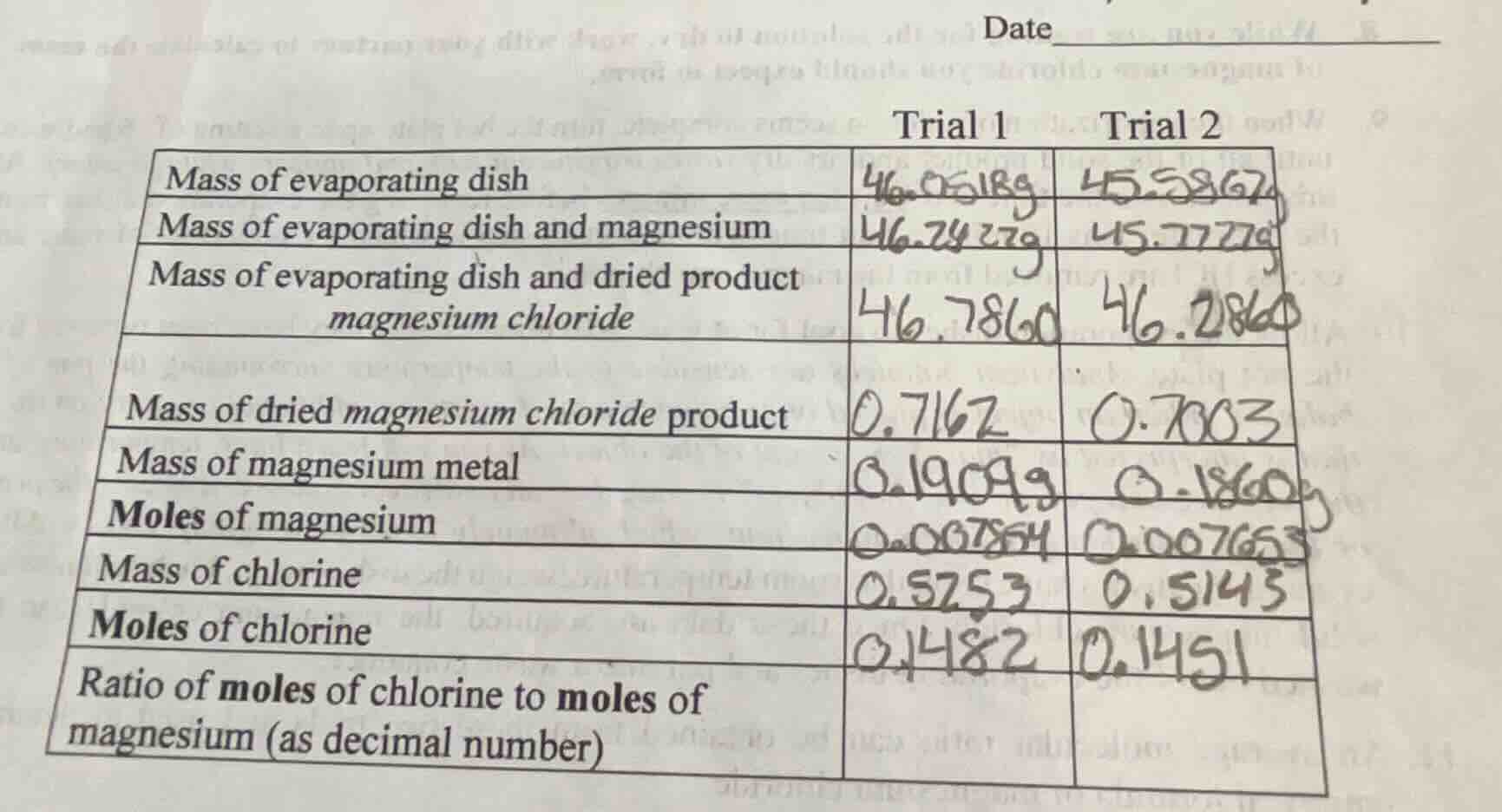
trial 1 trial 2
mass of evaporating dish 46.0618g 45.5867g
mass of evaporating dish and magnesium 46.2427g 45.7727g
mass of evaporating dish and dried product magnesium chloride 46.7860 46.2860
mass of dried magnesium chloride product 0.7162 0.7003
mass of magnesium metal 0.1909g 0.1860g
moles of magnesium 0.007864 0.007653
mass of chlorine 0.5253 0.5143
moles of chlorine 0.1482 0.1451
ratio of moles of chlorine to moles of magnesium (as decimal number)
Step1: Calculate ratio for Trial 1
Divide moles of Cl by moles of Mg: $\frac{0.1482}{0.007864}$
Step2: Calculate ratio for Trial 2
Divide moles of Cl by moles of Mg: $\frac{0.1451}{0.007653}$
Step3: Round to reasonable decimals
Round results to 2 decimal places
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Trial 1: 18.84
Trial 2: 18.96