QUESTION IMAGE
Question
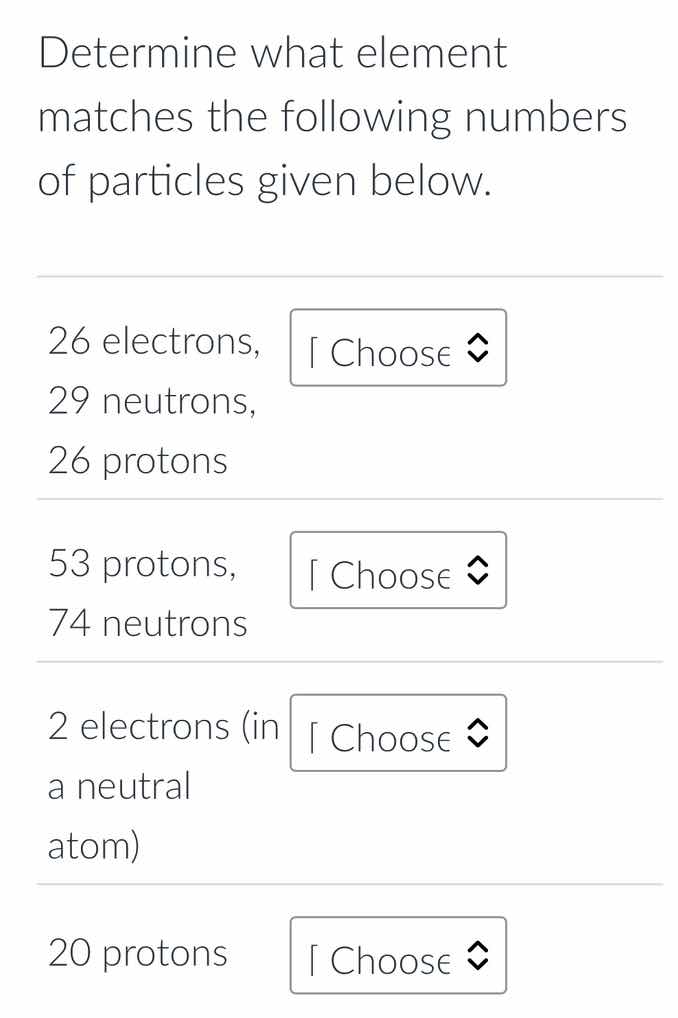
determine what element matches the following numbers of particles given below.
26 electrons,
29 neutrons,
26 protons
53 protons,
74 neutrons
2 electrons (in a neutral atom)
20 protons
Brief Explanations
- For a neutral atom, the number of protons equals the atomic number, which defines the element. The number of neutrons gives the isotope, but the element is identified by protons.
- For the first set: 26 protons = atomic number 26, which is Iron (Fe).
- For the second set: 53 protons = atomic number 53, which is Iodine (I).
- For the third set: 2 electrons in a neutral atom means 2 protons, atomic number 2, which is Helium (He).
- For the fourth set: 20 protons = atomic number 20, which is Calcium (Ca).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 26 electrons, 29 neutrons, 26 protons: Iron (Fe)
- 53 protons, 74 neutrons: Iodine (I)
- 2 electrons (in a neutral atom): Helium (He)
- 20 protons: Calcium (Ca)