QUESTION IMAGE
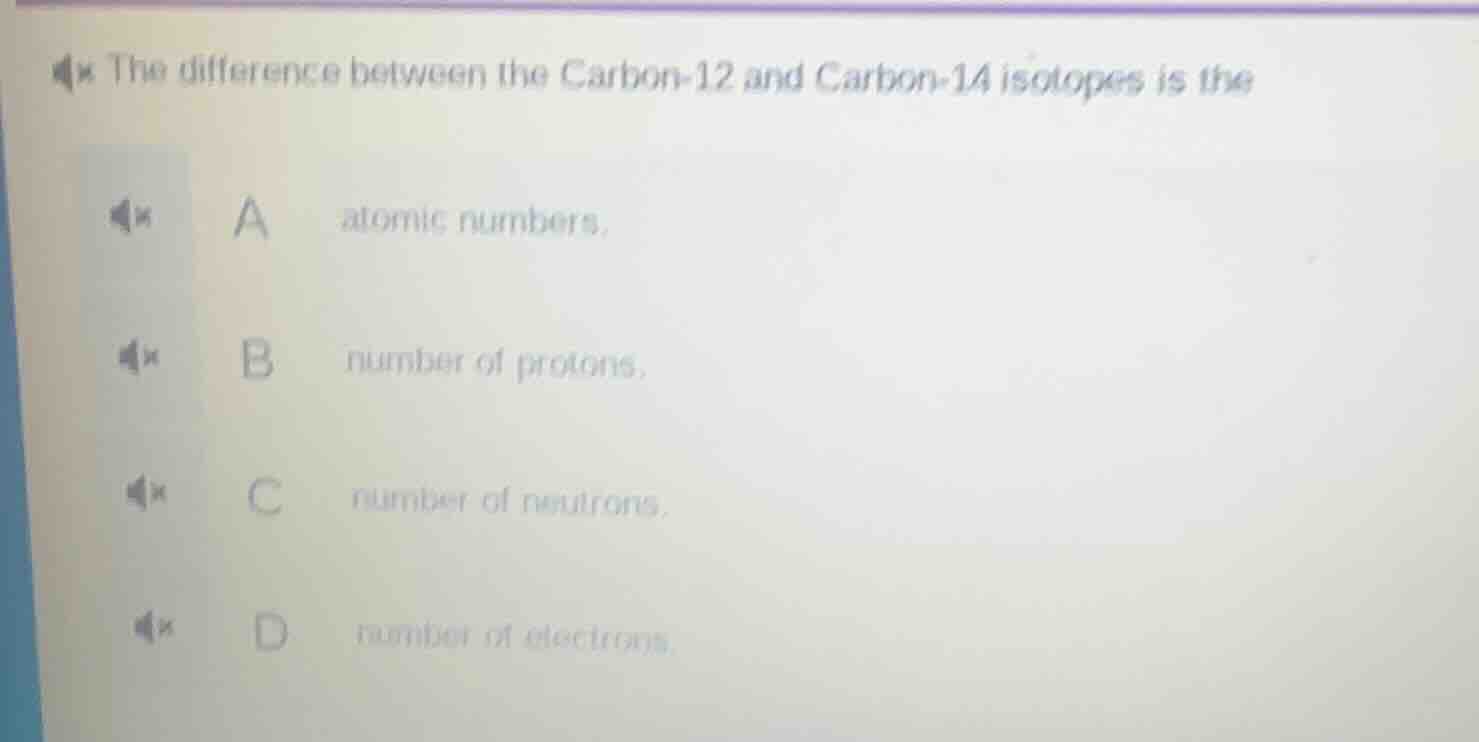
Question
the difference between the carbon-12 and carbon-14 isotopes is the a atomic numbers. b number of protons. c number of neutrons. d number of electrons.
Isotopes of an element have the same atomic number (equal to number of protons and electrons in neutral atoms), but differ in mass number. The mass number is the sum of protons and neutrons. Carbon has an atomic number of 6, so both C-12 and C-14 have 6 protons and 6 electrons. For C-12, neutrons = 12 - 6 = 6; for C-14, neutrons = 14 - 6 = 8. Thus, they differ in neutron count.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. number of neutrons.