QUESTION IMAGE
Question
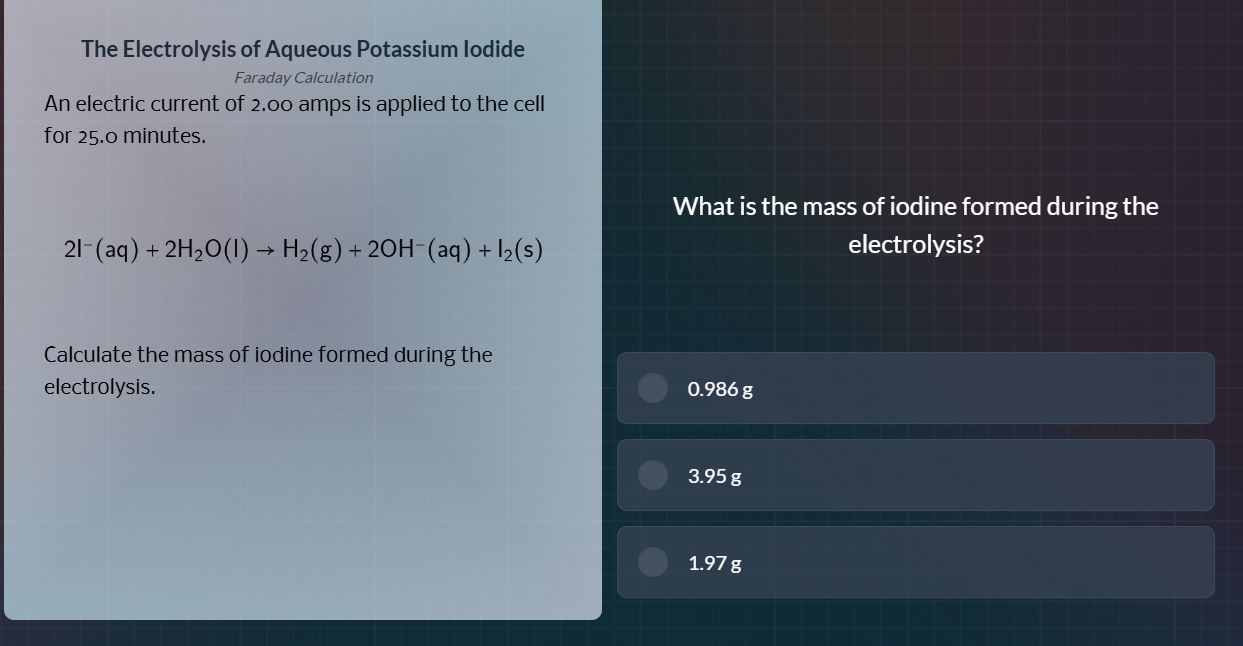
the electrolysis of aqueous potassium iodide
faraday calculation
an electric current of 2.00 amps is applied to the cell
for 25.0 minutes.
\\( 2\text{i}^-(\text{aq}) + 2\text{h}_2\text{o}(\text{l}) \
ightarrow \text{h}_2(\text{g}) + 2\text{oh}^-(\text{aq}) + \text{i}_2(\text{s}) \\)
calculate the mass of iodine formed during the
electrolysis.
what is the mass of iodine formed during the
electrolysis?
0.986 g
3.95 g
1.97 g
Step1: Calculate the charge (Q)
The formula for charge is \( Q = I \times t \). The current \( I = 2.00 \, \text{A} \) and time \( t = 25.0 \, \text{minutes} = 25.0 \times 60 = 1500 \, \text{s} \). So, \( Q = 2.00 \, \text{A} \times 1500 \, \text{s} = 3000 \, \text{C} \).
Step2: Find moles of electrons (n)
Using Faraday's constant \( F = 96485 \, \text{C/mol e}^- \), moles of electrons \( n = \frac{Q}{F} = \frac{3000 \, \text{C}}{96485 \, \text{C/mol e}^-} \approx 0.0311 \, \text{mol e}^- \).
Step3: Relate moles of electrons to moles of \( I_2 \)
From the reaction \( 2\text{I}^-
ightarrow \text{I}_2 + 2\text{e}^- \), 2 moles of electrons produce 1 mole of \( \text{I}_2 \). So, moles of \( \text{I}_2 = \frac{0.0311 \, \text{mol e}^-}{2} \approx 0.01555 \, \text{mol} \).
Step4: Calculate mass of \( \text{I}_2 \)
Molar mass of \( \text{I}_2 = 2 \times 126.90 \, \text{g/mol} = 253.80 \, \text{g/mol} \). Mass = moles × molar mass = \( 0.01555 \, \text{mol} \times 253.80 \, \text{g/mol} \approx 3.95 \, \text{g} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3.95 g