QUESTION IMAGE
Question
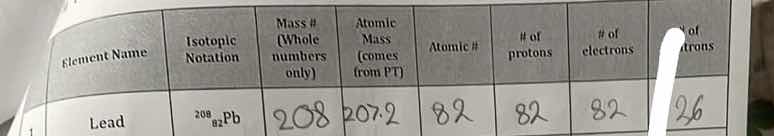
element name,isotopic notation,mass # (whole numbers only),atomic mass (comes from pt),atomic #,# of protons,# of electrons,# of neutrons; lead,\ce{^{208}_{82}pb},208,207.2,82,82,82,26
Step 1: Recall Isotope Basics
For an isotope \( ^{A}_{Z}\text{X} \), \( A \) is mass number (protons + neutrons), \( Z \) is atomic number (protons = electrons in neutral atom), atomic mass is from periodic table, neutrons = \( A - Z \).
Step 2: Analyze Lead Isotope \( ^{208}_{82}\text{Pb} \)
- Mass #: Given as \( 208 \) (whole number from notation).
- Atomic Mass: From PT, lead (Pb) has atomic mass \( 207.2 \).
- Atomic # (\( Z \)): From notation, \( Z = 82 \) (bottom number).
- # of protons: \( = Z = 82 \) (protons = atomic number).
- # of electrons: In neutral atom, electrons = protons = \( 82 \).
- # of neutrons: \( = \text{Mass #} - \text{Atomic #} = 208 - 82 = 126 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Element Name | Isotopic Notation | Mass # | Atomic Mass | Atomic # | # of protons | # of electrons | # of neutrons |
|---|
(Note: The original "26" for neutrons was incorrect; correct calculation gives 126.)