QUESTION IMAGE
Question
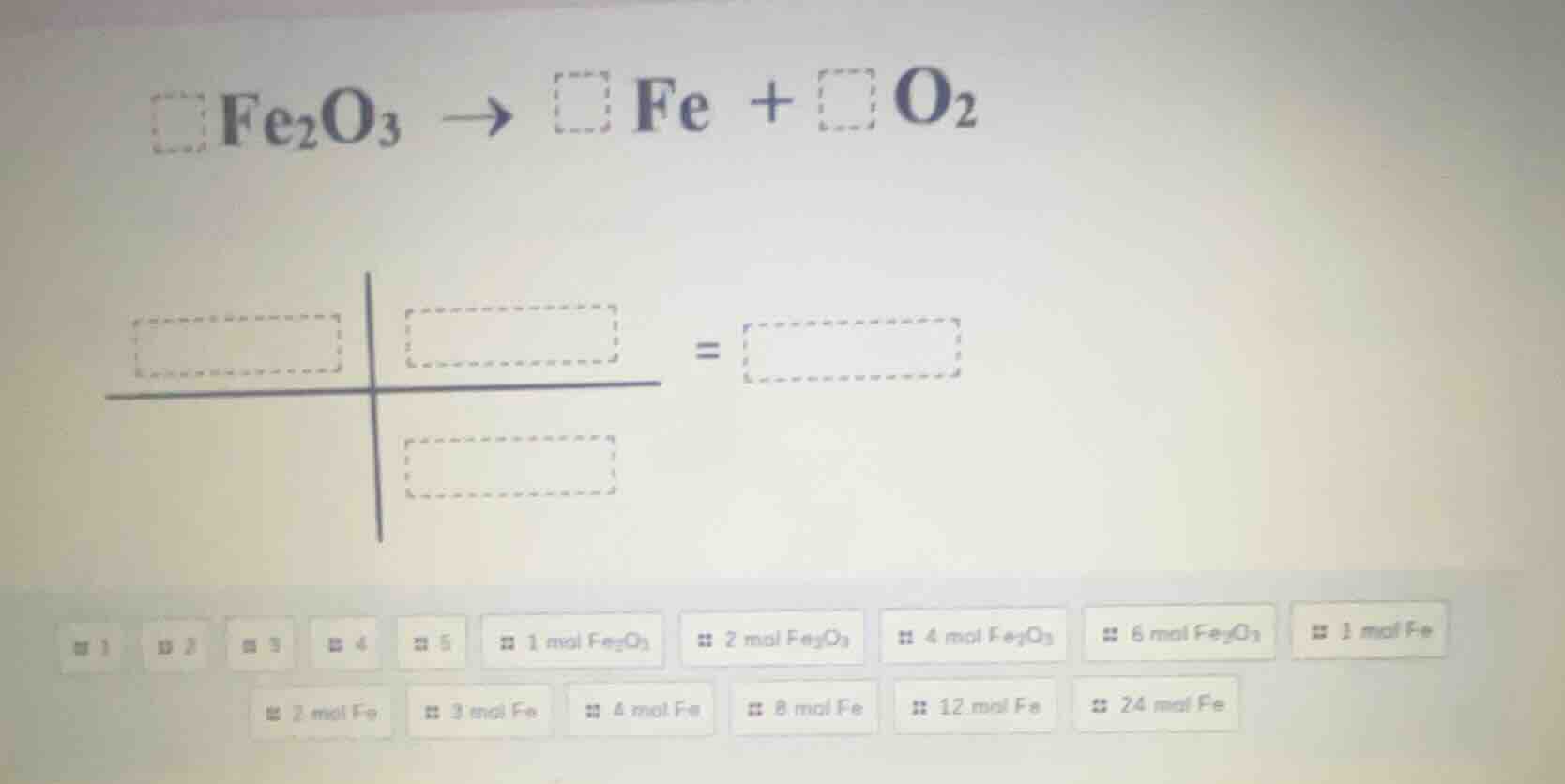
fe₂o₃ → fe + o₂
Step1: Balance the chemical equation
The unbalanced equation is $\ce{Fe_{2}O_{3} -> Fe + O_{2}}$. First, balance the iron (Fe) atoms. There are 2 Fe atoms in $\ce{Fe_{2}O_{3}}$, so we put a coefficient of 2 in front of Fe: $\ce{Fe_{2}O_{3} -> 2Fe + O_{2}}$. Now balance the oxygen (O) atoms. There are 3 O atoms in $\ce{Fe_{2}O_{3}}$, and $\ce{O_{2}}$ has 2 O atoms. The least common multiple of 3 and 2 is 6. So we put a coefficient of 2 in front of $\ce{Fe_{2}O_{3}}$ (to get 6 O atoms) and a coefficient of 3 in front of $\ce{O_{2}}$ (also 6 O atoms). Then adjust the Fe atoms: with 2 $\ce{Fe_{2}O_{3}}$, we have 4 Fe atoms, so the balanced equation is $\ce{2Fe_{2}O_{3} -> 4Fe + 3O_{2}}$.
Step2: Determine the mole ratios
From the balanced equation $\ce{2Fe_{2}O_{3} -> 4Fe + 3O_{2}}$, the mole ratio of $\ce{Fe_{2}O_{3}}$ to Fe is $2:4$ (or simplified $1:2$), and the mole ratio of $\ce{Fe_{2}O_{3}}$ to $\ce{O_{2}}$ is $2:3$.
If we take 2 mol of $\ce{Fe_{2}O_{3}}$, then:
- Moles of Fe: Using the ratio $2\ \text{mol}\ \ce{Fe_{2}O_{3}} : 4\ \text{mol}\ \text{Fe}$, so 4 mol of Fe.
- Moles of $\ce{O_{2}}$: Using the ratio $2\ \text{mol}\ \ce{Fe_{2}O_{3}} : 3\ \text{mol}\ \ce{O_{2}}$, so 3 mol of $\ce{O_{2}}$.
(Assuming we are matching the moles based on the balanced equation, for example, if we have 2 mol of $\ce{Fe_{2}O_{3}}$, we get 4 mol of Fe and 3 mol of $\ce{O_{2}}$)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The balanced equation is $\ce{2Fe_{2}O_{3} -> 4Fe + 3O_{2}}$, so the coefficients are 2 (for $\ce{Fe_{2}O_{3}}$), 4 (for Fe), and 3 (for $\ce{O_{2}}$). If using moles, for example, 2 mol of $\ce{Fe_{2}O_{3}}$ produces 4 mol of Fe and 3 mol of $\ce{O_{2}}$.