QUESTION IMAGE
Question
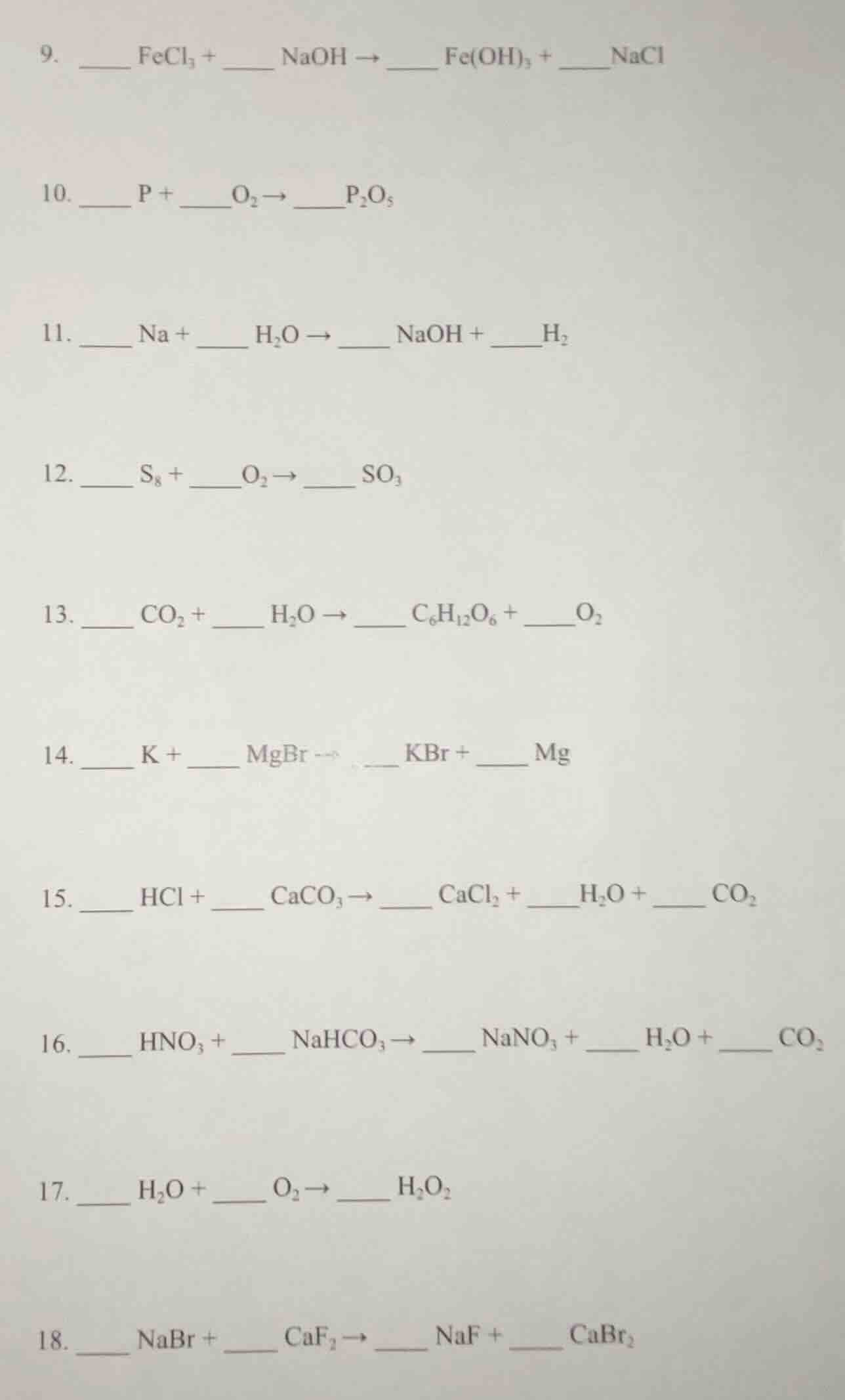
- \underline{\quad\quad} fecl₃ + \underline{\quad\quad} naoh \
ightarrow \underline{\quad\quad} fe(oh)₃ + \underline{\quad\quad}nacl\
\
- \underline{\quad\quad} p + \underline{\quad\quad}o₂ \
ightarrow \underline{\quad\quad}p₂o₅\
\
- \underline{\quad\quad} na + \underline{\quad\quad} h₂o \
ightarrow \underline{\quad\quad} naoh + \underline{\quad\quad}h₂\
\
- \underline{\quad\quad} s₈ + \underline{\quad\quad}o₂ \
ightarrow \underline{\quad\quad} so₃\
\
- \underline{\quad\quad} co₂ + \underline{\quad\quad} h₂o \
ightarrow \underline{\quad\quad} c₆h₁₂o₆ + \underline{\quad\quad}o₂\
\
- \underline{\quad\quad} k + \underline{\quad\quad} mgbr \
ightarrow \underline{\quad\quad} kbr + \underline{\quad\quad} mg\
\
- \underline{\quad\quad} hcl + \underline{\quad\quad} caco₃ \
ightarrow \underline{\quad\quad} cacl₂ + \underline{\quad\quad}h₂o + \underline{\quad\quad} co₂\
\
- \underline{\quad\quad} hno₃ + \underline{\quad\quad} nahco₃ \
ightarrow \underline{\quad\quad} nano₃ + \underline{\quad\quad} h₂o + \underline{\quad\quad} co₂\
\
- \underline{\quad\quad} h₂o + \underline{\quad\quad} o₂ \
ightarrow \underline{\quad\quad} h₂o₂\
\
- \underline{\quad\quad} nabr + \underline{\quad\quad} caf₂ \
ightarrow \underline{\quad\quad} naf + \underline{\quad\quad} cabr₂
Problem 9
Step1: Balance Fe
There is 1 Fe in \( \ce{FeCl3} \) and 1 Fe in \( \ce{Fe(OH)3} \), so Fe is balanced.
Step2: Balance Cl
There are 3 Cl in \( \ce{FeCl3} \) and 1 Cl in \( \ce{NaCl} \). So we need 3 \( \ce{NaCl} \), which gives 3 Na.
Step3: Balance Na
There are 3 Na in \( \ce{NaCl} \), so we need 3 \( \ce{NaOH} \), which gives 3 OH.
Step4: Balance OH
There are 3 OH in \( \ce{NaOH} \) and 3 OH in \( \ce{Fe(OH)3} \), so OH is balanced.
So the balanced equation is \( \ce{1 FeCl3 + 3 NaOH -> 1 Fe(OH)3 + 3 NaCl} \)
Step1: Balance P
There are 2 P in \( \ce{P2O5} \), so we need 2 P on the left, so coefficient of P is 2.
Step2: Balance O
There are 5 O in \( \ce{P2O5} \) and 2 O in \( \ce{O2} \). The least common multiple of 5 and 2 is 10. So we need 5 \( \ce{O2} \) (10 O) and 2 \( \ce{P2O5} \) (10 O). But wait, if we have 2 \( \ce{P2O5} \), then P would be 4. So let's adjust. Let's balance O first. The number of O in \( \ce{P2O5} \) is 5, so coefficient of \( \ce{O2} \) is 5/2? No, we need whole numbers. So multiply all coefficients by 2. So P becomes 4, \( \ce{O2} \) becomes 5, and \( \ce{P2O5} \) becomes 2. Wait, no, original: \( \ce{P + O2 -> P2O5} \). Let's do it properly. Let coefficient of P be x, \( \ce{O2} \) be y, \( \ce{P2O5} \) be z. So x P, 2y O, 2z P and 5z O. So x = 2z (P balance), 2y = 5z (O balance). Let z = 2, then x = 4, y = 5. So the balanced equation is \( \ce{4 P + 5 O2 -> 2 P2O5} \)
Step1: Balance H
There are 2 H in \( \ce{H2} \) and 1 H in \( \ce{NaOH} \). Let's balance H first. Let coefficient of \( \ce{H2} \) be 1, then we need 2 \( \ce{NaOH} \) (2 H), which gives 2 Na.
Step2: Balance Na
There are 2 Na in \( \ce{NaOH} \), so coefficient of Na is 2.
Step3: Balance O
There are 2 O in \( \ce{NaOH} \) and 2 O in \( \ce{H2O} \) (since coefficient of \( \ce{H2O} \) is 2).
Step4: Balance H (again)
There are 2 \( \ce{H2O} \) (4 H) and 2 \( \ce{NaOH} \) (2 H) and 1 \( \ce{H2} \) (2 H). Wait, 4 H from \( \ce{H2O} \), 2 H from \( \ce{NaOH} \)? No, wait, the equation is \( \ce{Na + H2O -> NaOH + H2} \). Let's do it with variables. Let Na be a, \( \ce{H2O} \) be b, \( \ce{NaOH} \) be c, \( \ce{H2} \) be d. So a Na, 2b H, b O, c Na, c O, c H, 2d H. So Na: a = c. O: b = c. H: 2b = c + 2d. Since a = c = b, substitute: 2b = b + 2d => b = 2d. Let d = 1, then b = 2, c = 2, a = 2. So the balanced equation is \( \ce{2 Na + 2 H2O -> 2 NaOH + 1 H2} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1, 3, 1, 3