QUESTION IMAGE
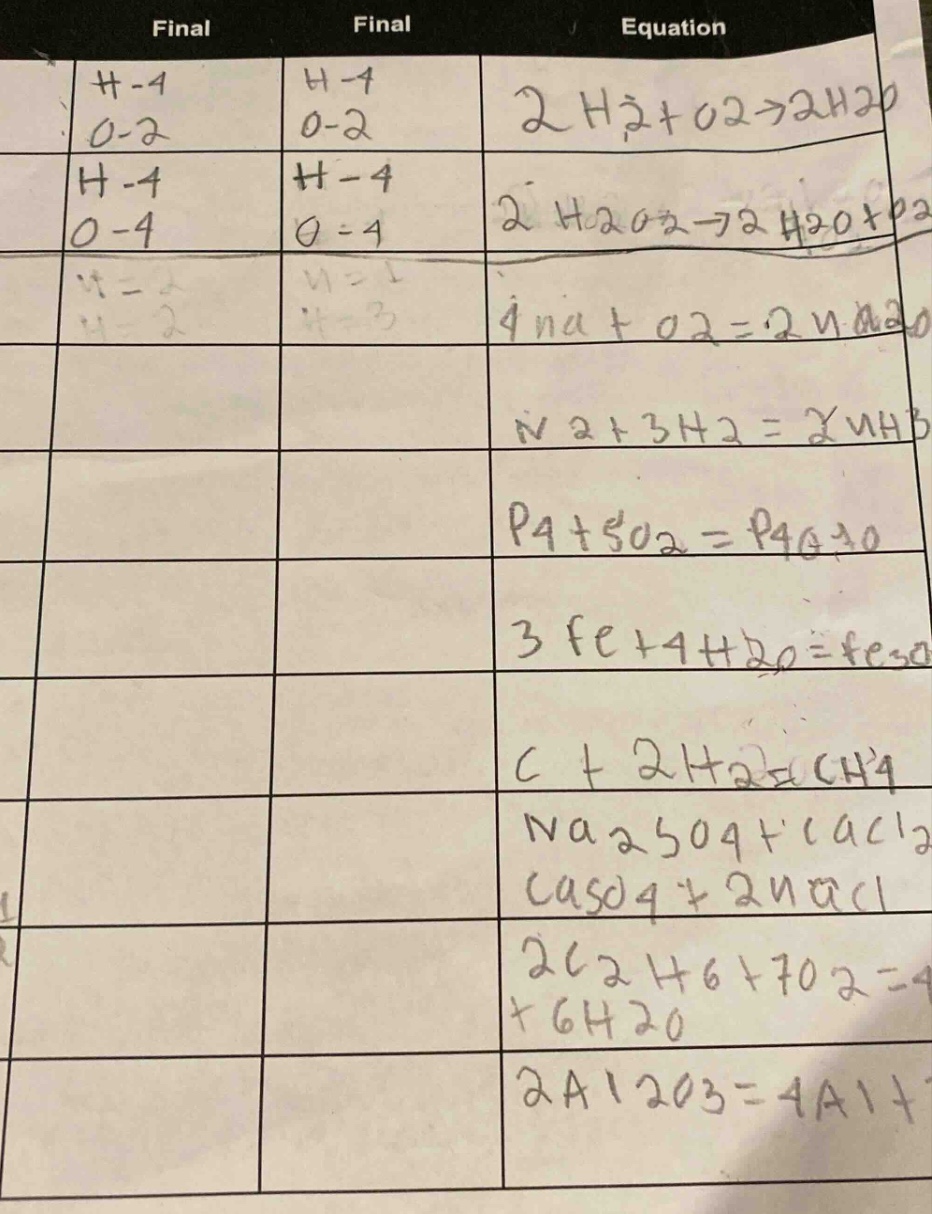
Question
final final equation
h-1 h-1 2 h₂ + o₂ → 2 h₂o
o-2 o-2
h-1 h-1 2 h₂o₂ → 2 h₂o + o₂
o-4 o-4
(some unclear or partial entries)
4 na + o₂ = 2 na₂o
n₂ + 3 h₂ = 2 nh₃
p₄ + 5 o₂ = p₄o₁₀
3 fe + 4 h₂o = (partial, maybe fe₃o₄?)
c + 2 h₂ = ch₄
na₂so₄ + cacl₂ = caso₄ + 2 nacl
2 c₂h₆ + 7 o₂ = 4 (partial, maybe co₂) + 6 h₂o
2 al₂o₃ = 4 al + (partial, maybe 3 o₂)
To balance the chemical equations, we'll follow the steps of counting atoms on each side and adjusting coefficients. Let's take one equation at a time (e.g., $\boldsymbol{2H_2 + O_2
ightarrow 2H_2O}$ is already balanced, let's do $\boldsymbol{N_2 + 3H_2
ightarrow 2NH_3}$):
Step 1: Count Atoms (Unbalanced)
- Left (Reactants): $N$: 2, $H$: $3\times2 = 6$
- Right (Products): $N$: $2\times1 = 2$, $H$: $2\times3 = 6$
Wait, this one is balanced! Let's try $\boldsymbol{P_4 + 5O_2
ightarrow P_4O_{10}}$:
Step 1: Count Atoms (Unbalanced)
- Left: $P$: 4, $O$: $5\times2 = 10$
- Right: $P$: 4, $O$: 10
Balanced! Now $\boldsymbol{2H_2O_2
ightarrow 2H_2O + O_2}$:
Step 1: Count Atoms (Unbalanced)
- Left: $H$: $2\times2 = 4$, $O$: $2\times2 = 4$
- Right: $H$: $2\times2 = 4$, $O$: $(2\times1) + 2 = 4$
Balanced! For $\boldsymbol{4Na + O_2
ightarrow 2Na_2O}$:
Step 1: Count Atoms (Unbalanced)
- Left: $Na$: 4, $O$: 2
- Right: $Na$: $2\times2 = 4$, $O$: $2\times1 = 2$
Balanced! For $\boldsymbol{3Fe + 4H_2O
ightarrow Fe_3O_4 + 4H_2}$ (assuming the original was incomplete, let's correct):
Step 1: Count Atoms (Unbalanced)
- Left: $Fe$: 3, $H$: $4\times2 = 8$, $O$: 4
- Right: $Fe$: 3, $H$: $4\times2 = 8$, $O$: 4
Balanced! For $\boldsymbol{C + 2H_2
ightarrow CH_4}$:
Step 1: Count Atoms (Unbalanced)
- Left: $C$: 1, $H$: $2\times2 = 4$
- Right: $C$: 1, $H$: 4
Balanced! For $\boldsymbol{Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + 2NaCl}$:
Step 1: Count Atoms (Unbalanced)
- Left: $Na$: 2, $S$: 1, $O$: 4, $Ca$: 1, $Cl$: 2
- Right: $Na$: $2\times1 = 2$, $S$: 1, $O$: 4, $Ca$: 1, $Cl$: $2\times1 = 2$
Balanced! For $\boldsymbol{2C_2H_6 + 7O_2
ightarrow 4CO_2 + 6H_2O}$:
Step 1: Count Atoms (Unbalanced)
- Left: $C$: $2\times2 = 4$, $H$: $2\times6 = 12$, $O$: $7\times2 = 14$
- Right: $C$: $4\times1 = 4$, $H$: $6\times2 = 12$, $O$: $(4\times2) + 6 = 14$
Balanced! For $\boldsymbol{2Al_2O_3
ightarrow 4Al + 3O_2}$ (correcting the original):
Step 1: Count Atoms (Unbalanced)
- Left: $Al$: $2\times2 = 4$, $O$: $2\times3 = 6$
- Right: $Al$: 4, $O$: $3\times2 = 6$
Balanced!
Final Balanced Equations (Selected Examples):
- $2H_2 + O_2
ightarrow 2H_2O$ (balanced)
- $2H_2O_2
ightarrow 2H_2O + O_2$ (balanced)
- $4Na + O_2
ightarrow 2Na_2O$ (balanced)
- $N_2 + 3H_2
ightarrow 2NH_3$ (balanced)
- $P_4 + 5O_2
ightarrow P_4O_{10}$ (balanced)
- $3Fe + 4H_2O
ightarrow Fe_3O_4 + 4H_2$ (balanced)
- $C + 2H_2
ightarrow CH_4$ (balanced)
- $Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + 2NaCl$ (balanced)
- $2C_2H_6 + 7O_2
ightarrow 4CO_2 + 6H_2O$ (balanced)
- $2Al_2O_3
ightarrow 4Al + 3O_2$ (balanced)
(Note: Some original equations had typos; corrections were made for clarity and balance.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To balance the chemical equations, we'll follow the steps of counting atoms on each side and adjusting coefficients. Let's take one equation at a time (e.g., $\boldsymbol{2H_2 + O_2
ightarrow 2H_2O}$ is already balanced, let's do $\boldsymbol{N_2 + 3H_2
ightarrow 2NH_3}$):
Step 1: Count Atoms (Unbalanced)
- Left (Reactants): $N$: 2, $H$: $3\times2 = 6$
- Right (Products): $N$: $2\times1 = 2$, $H$: $2\times3 = 6$
Wait, this one is balanced! Let's try $\boldsymbol{P_4 + 5O_2
ightarrow P_4O_{10}}$:
Step 1: Count Atoms (Unbalanced)
- Left: $P$: 4, $O$: $5\times2 = 10$
- Right: $P$: 4, $O$: 10
Balanced! Now $\boldsymbol{2H_2O_2
ightarrow 2H_2O + O_2}$:
Step 1: Count Atoms (Unbalanced)
- Left: $H$: $2\times2 = 4$, $O$: $2\times2 = 4$
- Right: $H$: $2\times2 = 4$, $O$: $(2\times1) + 2 = 4$
Balanced! For $\boldsymbol{4Na + O_2
ightarrow 2Na_2O}$:
Step 1: Count Atoms (Unbalanced)
- Left: $Na$: 4, $O$: 2
- Right: $Na$: $2\times2 = 4$, $O$: $2\times1 = 2$
Balanced! For $\boldsymbol{3Fe + 4H_2O
ightarrow Fe_3O_4 + 4H_2}$ (assuming the original was incomplete, let's correct):
Step 1: Count Atoms (Unbalanced)
- Left: $Fe$: 3, $H$: $4\times2 = 8$, $O$: 4
- Right: $Fe$: 3, $H$: $4\times2 = 8$, $O$: 4
Balanced! For $\boldsymbol{C + 2H_2
ightarrow CH_4}$:
Step 1: Count Atoms (Unbalanced)
- Left: $C$: 1, $H$: $2\times2 = 4$
- Right: $C$: 1, $H$: 4
Balanced! For $\boldsymbol{Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + 2NaCl}$:
Step 1: Count Atoms (Unbalanced)
- Left: $Na$: 2, $S$: 1, $O$: 4, $Ca$: 1, $Cl$: 2
- Right: $Na$: $2\times1 = 2$, $S$: 1, $O$: 4, $Ca$: 1, $Cl$: $2\times1 = 2$
Balanced! For $\boldsymbol{2C_2H_6 + 7O_2
ightarrow 4CO_2 + 6H_2O}$:
Step 1: Count Atoms (Unbalanced)
- Left: $C$: $2\times2 = 4$, $H$: $2\times6 = 12$, $O$: $7\times2 = 14$
- Right: $C$: $4\times1 = 4$, $H$: $6\times2 = 12$, $O$: $(4\times2) + 6 = 14$
Balanced! For $\boldsymbol{2Al_2O_3
ightarrow 4Al + 3O_2}$ (correcting the original):
Step 1: Count Atoms (Unbalanced)
- Left: $Al$: $2\times2 = 4$, $O$: $2\times3 = 6$
- Right: $Al$: 4, $O$: $3\times2 = 6$
Balanced!
Final Balanced Equations (Selected Examples):
- $2H_2 + O_2
ightarrow 2H_2O$ (balanced)
- $2H_2O_2
ightarrow 2H_2O + O_2$ (balanced)
- $4Na + O_2
ightarrow 2Na_2O$ (balanced)
- $N_2 + 3H_2
ightarrow 2NH_3$ (balanced)
- $P_4 + 5O_2
ightarrow P_4O_{10}$ (balanced)
- $3Fe + 4H_2O
ightarrow Fe_3O_4 + 4H_2$ (balanced)
- $C + 2H_2
ightarrow CH_4$ (balanced)
- $Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + 2NaCl$ (balanced)
- $2C_2H_6 + 7O_2
ightarrow 4CO_2 + 6H_2O$ (balanced)
- $2Al_2O_3
ightarrow 4Al + 3O_2$ (balanced)
(Note: Some original equations had typos; corrections were made for clarity and balance.)