QUESTION IMAGE
Question
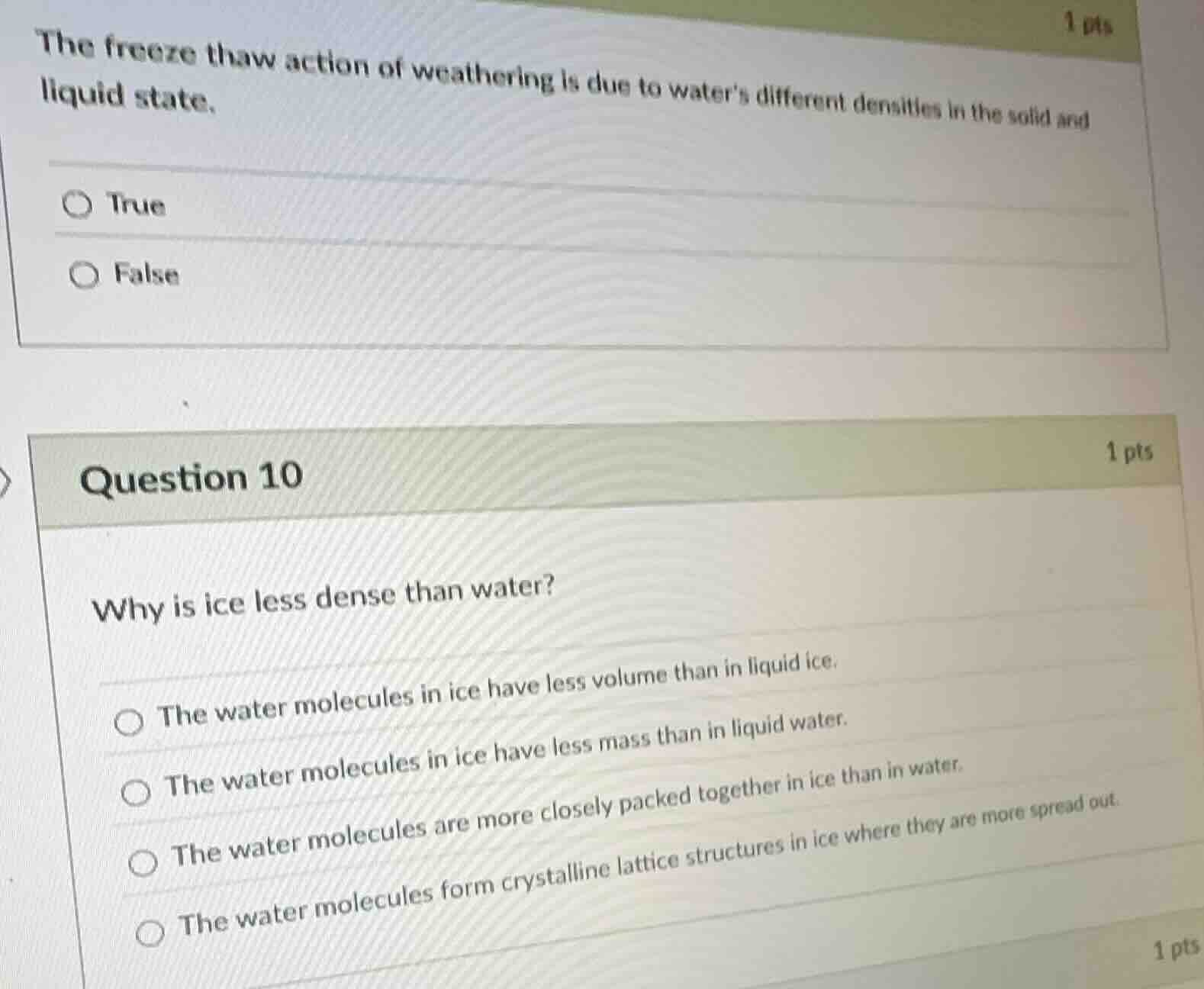
the freeze thaw action of weathering is due to waters different densities in the solid and liquid state. true false question 10 1 pts why is ice less dense than water? the water molecules in ice have less volume than in liquid ice. the water molecules in ice have less mass than in liquid water. the water molecules are more closely packed together in ice than in water. the water molecules form crystalline lattice structures in ice where they are more spread out.
First Question (Freeze - thaw action)
Freeze - thaw weathering (also known as frost shattering) occurs because water expands when it freezes (due to ice being less dense than liquid water). When water in cracks freezes, it expands, exerting pressure on the rock. When it thaws, the pressure is released. The difference in density (ice is less dense than liquid water) leads to expansion on freezing, which drives this weathering process. So the statement is True.
- Option 1: Incorrect. Ice has more volume than liquid water (since it's less dense), and molecules don't have "less volume" in ice. Also, "liquid ice" is not a correct term (it's liquid water).
- Option 2: Incorrect. The mass of water molecules remains the same whether in ice or liquid water (conservation of mass, same number of molecules).
- Option 3: Incorrect. Water molecules are more closely packed in liquid water than in ice.
- Option 4: Correct. In ice, water molecules form a crystalline lattice structure (hexagonal arrangement) where the molecules are more spread out compared to the more random, closely - packed arrangement in liquid water. This increased spacing in the lattice makes ice less dense.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
True