QUESTION IMAGE
Question
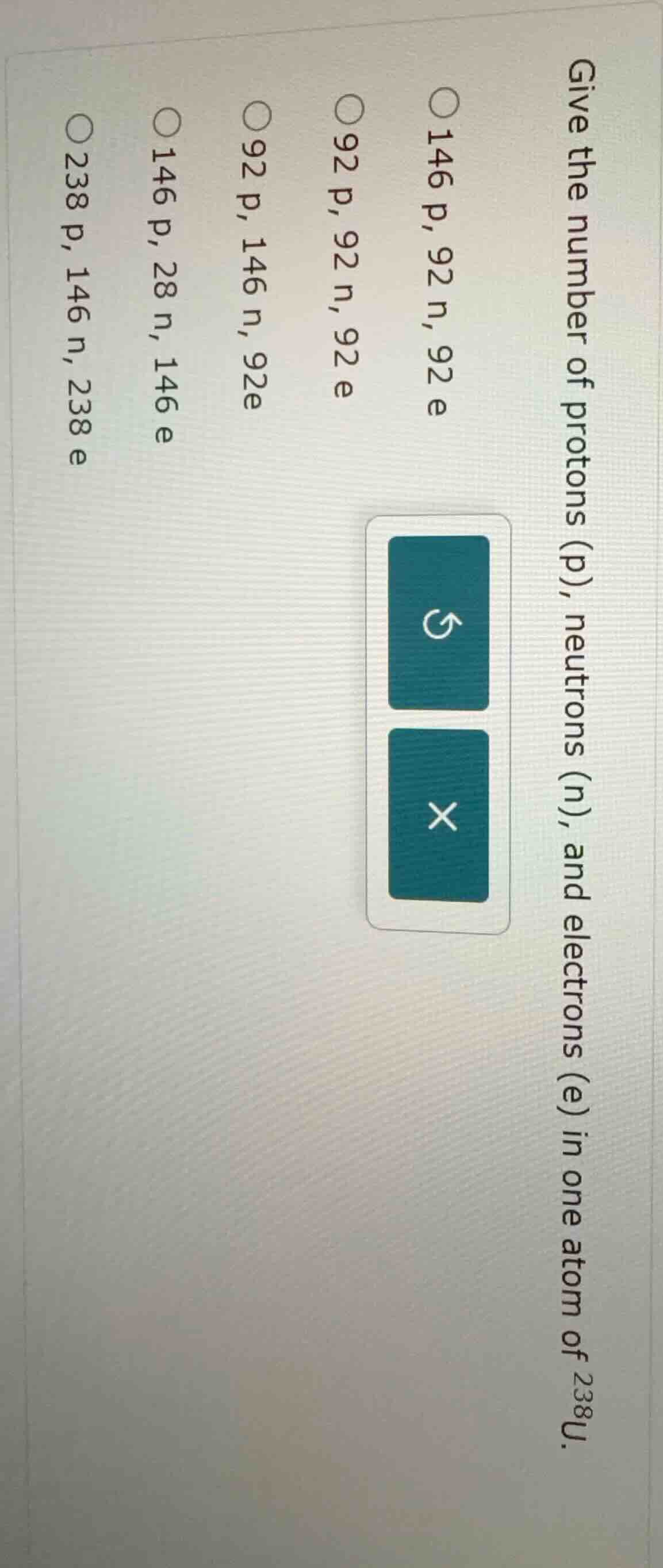
give the number of protons (p), neutrons (n), and electrons (e) in one atom of (^{238}\text{u}).
○ 146 p, 92 n, 92 e
○ 92 p, 92 n, 92 e
○ 92 p, 146 n, 92 e
○ 146 p, 28 n, 146 e
○ 238 p, 146 n, 238 e
Step1: Determine number of protons
For an atom \( ^{A}_{Z}X \), the atomic number \( Z \) is the number of protons. For \( ^{238}_{92}\text{U} \), \( Z = 92 \), so protons (\( p \)) = 92.
Step2: Determine number of electrons
In a neutral atom, number of electrons (\( e \)) equals number of protons. So \( e = 92 \).
Step3: Determine number of neutrons
Mass number \( A = 238 \), neutrons (\( n \)) = \( A - Z = 238 - 92 = 146 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
92 p, 146 n, 92 e (corresponding to the option "92 p, 146 n, 92 e")