QUESTION IMAGE
Question
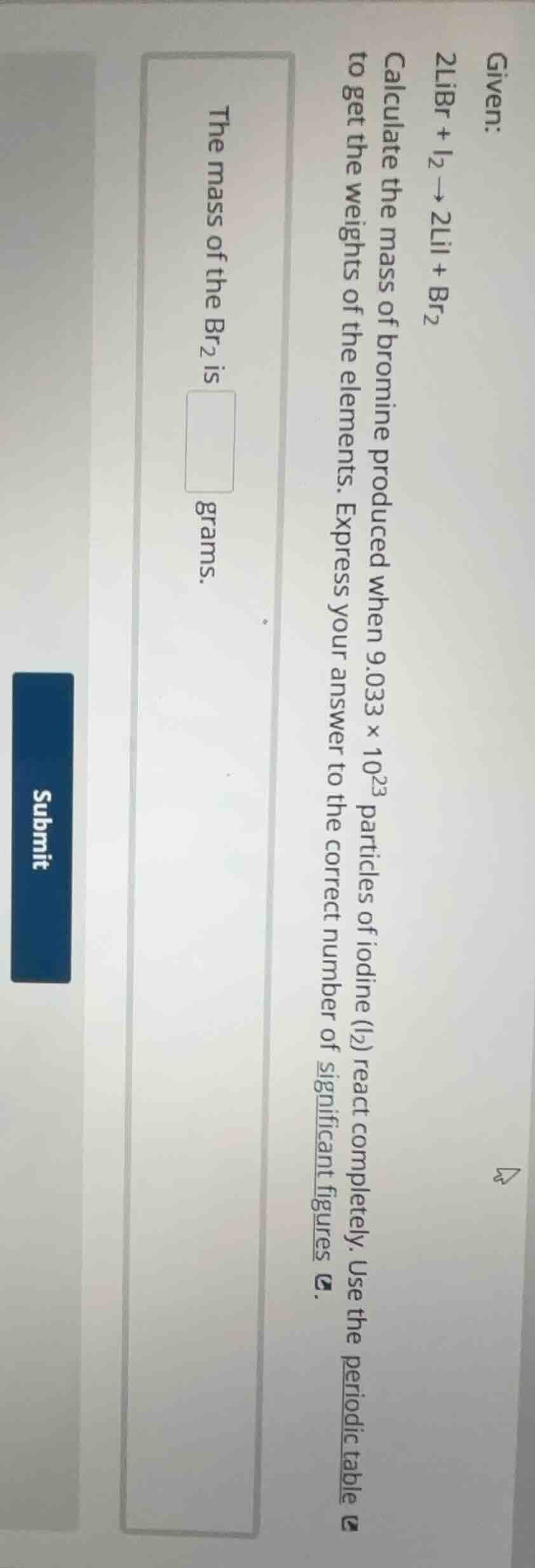
given: 2libr + i₂ → 2lii + br₂ calculate the mass of bromine produced when 9.033 × 10²³ particles of iodine (i₂) react completely. use the periodic table to get the weights of the elements. express your answer to the correct number of significant figures. the mass of the br₂ is \boxed{} grams. submit
Step1: Convert particles to moles
Use Avogadro's number ($N_A = 6.022 \times 10^{23} \text{ particles/mol}$)
$$n_{I_2} = \frac{9.033 \times 10^{23} \text{ particles}}{6.022 \times 10^{23} \text{ particles/mol}} = 1.500 \text{ mol}$$
Step2: Relate moles of $I_2$ to $Br_2$
From balanced equation, mole ratio $I_2:Br_2 = 1:1$
$$n_{Br_2} = n_{I_2} = 1.500 \text{ mol}$$
Step3: Calculate molar mass of $Br_2$
Molar mass of $Br = 79.90 \text{ g/mol}$, so:
$$M_{Br_2} = 2 \times 79.90 \text{ g/mol} = 159.8 \text{ g/mol}$$
Step4: Calculate mass of $Br_2$
Use $m = n \times M$
$$m_{Br_2} = 1.500 \text{ mol} \times 159.8 \text{ g/mol}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
239.7 grams