QUESTION IMAGE
Question
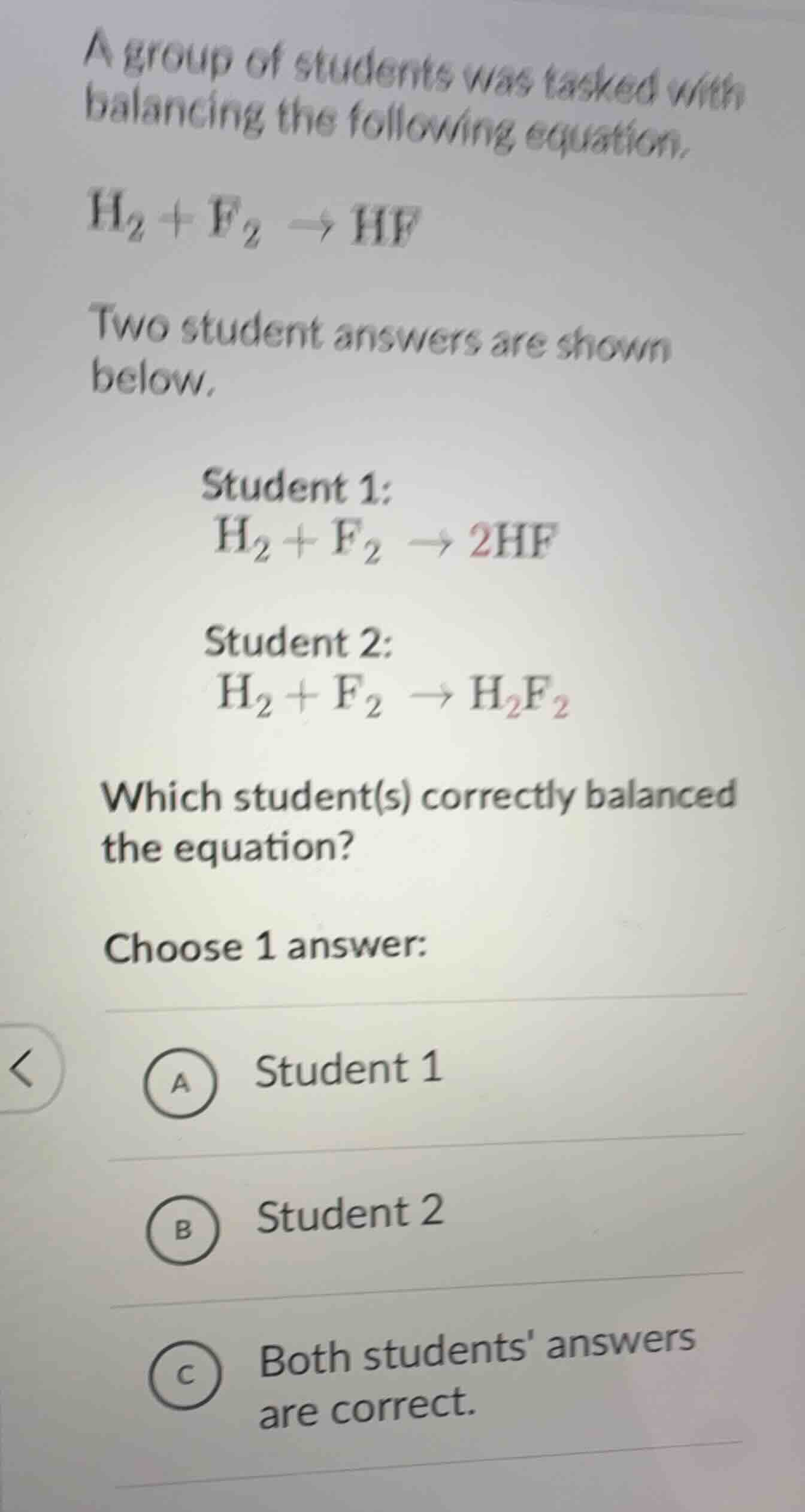
a group of students was tasked with balancing the following equation.\\(\ce{h_{2} + f_{2} \
ightarrow hf}\\)\
two student answers are shown below.\
\
student 1:\
\\(\ce{h_{2} + f_{2} \
ightarrow 2hf}\\)\
\
student 2:\
\\(\ce{h_{2} + f_{2} \
ightarrow h_{2}f_{2}}\\)\
\
which student(s) correctly balanced the equation?\
choose 1 answer:\
a student 1\
b student 2\
c both students answers are correct.
To balance a chemical equation, the number of each type of atom must be the same on both sides. For the reaction \( \ce{H_2 + F_2
ightarrow HF} \):
- Student 1: The equation is \( \ce{H_2 + F_2
ightarrow 2HF} \). On the left, there are 2 H atoms (from \( \ce{H_2} \)) and 2 F atoms (from \( \ce{F_2} \)). On the right, \( 2\ce{HF} \) has \( 2 \times 1 = 2 \) H atoms and \( 2 \times 1 = 2 \) F atoms. So, the number of H and F atoms is balanced.
- Student 2: The equation is \( \ce{H_2 + F_2
ightarrow H_2F_2} \). On the left, 2 H and 2 F atoms. On the right, \( \ce{H_2F_2} \) has 2 H and 2 F atoms. This also balances the atoms (though \( \ce{H_2F_2} \) is not a typical way to write HF, the atom count is balanced).
Since both students have equal numbers of H and F atoms on both sides, both answers are correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Both students' answers are correct.