QUESTION IMAGE
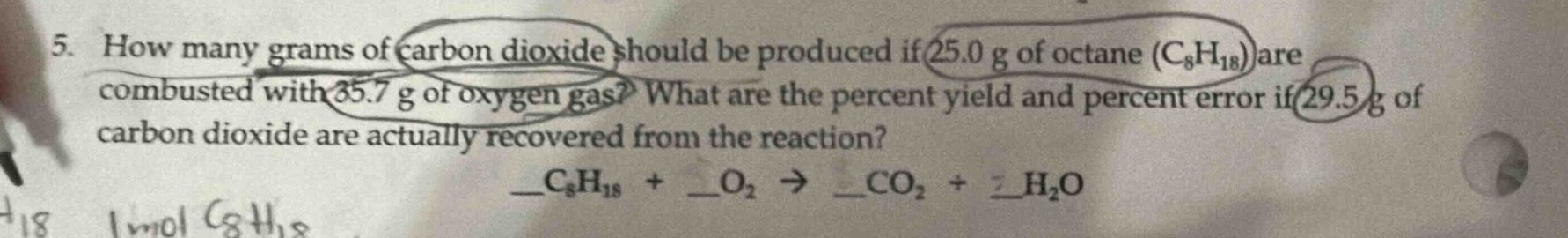
Question
- how many grams of carbon dioxide should be produced if 25.0 g of octane ($\text{c}_8\text{h}_{18}$) are combusted with 35.7 g of oxygen gas? what are the percent yield and percent error if 29.5 g of carbon dioxide are actually recovered from the reaction?
$\underline{\quad}\text{c}_8\text{h}_{18} + \underline{\quad}\text{o}_2 \
ightarrow \underline{\quad}\text{co}_2 + \underline{\quad}\text{h}_2\text{o}$
Step1: Balance the combustion equation
Balance the atoms for the reaction:
$$\ce{2C8H18 + 25O2 -> 16CO2 + 18H2O}$$
Step2: Calculate molar masses
Molar mass of $\ce{C8H18}$: $8\times12.01 + 18\times1.008 = 114.23\ \text{g/mol}$
Molar mass of $\ce{O2}$: $2\times16.00 = 32.00\ \text{g/mol}$
Molar mass of $\ce{CO2}$: $12.01 + 2\times16.00 = 44.01\ \text{g/mol}$
Step3: Find moles of reactants
Moles of $\ce{C8H18}$: $\frac{25.0\ \text{g}}{114.23\ \text{g/mol}} = 0.2189\ \text{mol}$
Moles of $\ce{O2}$: $\frac{35.7\ \text{g}}{32.00\ \text{g/mol}} = 1.1156\ \text{mol}$
Step4: Identify limiting reactant
From balanced equation, $\ce{C8H18:O2}$ mole ratio = $2:25$.
Required $\ce{O2}$ for 0.2189 mol $\ce{C8H18}$: $0.2189\ \text{mol} \times \frac{25}{2} = 2.736\ \text{mol}$
Available $\ce{O2}$ (1.1156 mol) < required, so $\ce{O2}$ is limiting.
Step5: Calculate theoretical yield of $\ce{CO2}$
From balanced equation, $\ce{O2:CO2}$ mole ratio = $25:16$.
Moles of $\ce{CO2}$: $1.1156\ \text{mol} \times \frac{16}{25} = 0.71398\ \text{mol}$
Theoretical mass: $0.71398\ \text{mol} \times 44.01\ \text{g/mol} = 31.42\ \text{g}$
Step6: Calculate percent yield
Percent yield = $\frac{\text{actual yield}}{\text{theoretical yield}} \times 100\%$
$$\frac{29.5\ \text{g}}{31.42\ \text{g}} \times 100\% = 93.9\%$$
Step7: Calculate percent error
Percent error = $\frac{|\text{actual - theoretical}|}{\text{theoretical yield}} \times 100\%$
$$\frac{|29.5 - 31.42|}{31.42} \times 100\% = 6.11\%$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Theoretical yield of $\ce{CO2}$: $31.4\ \text{g}$
Percent yield: $93.9\%$
Percent error: $6.11\%$