QUESTION IMAGE
Question
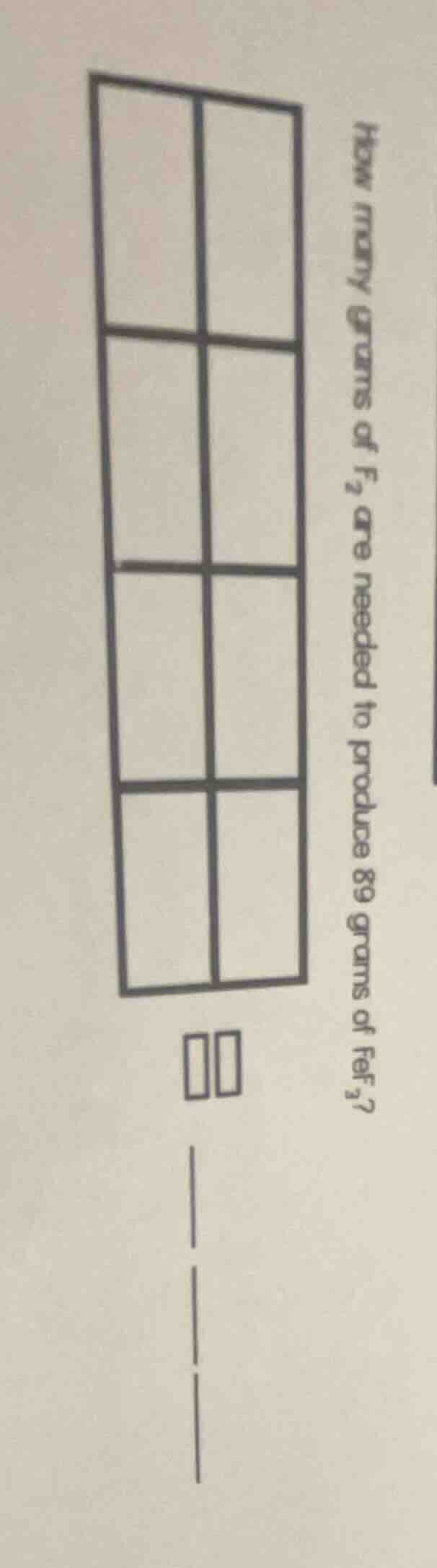
how many grams of f₂ are needed to produce 89 grams of fef₃?
First, we need to know the chemical formula and molar masses. The reaction to form \( \text{FeF}_3 \) from \( \text{Fe} \) and \( \text{F}_2 \) is: \( 2\text{Fe} + 3\text{F}_2
ightarrow 2\text{FeF}_3 \)
Step 1: Calculate molar mass of \( \text{FeF}_3 \)
Molar mass of \( \text{Fe} = 55.85 \, \text{g/mol} \), molar mass of \( \text{F} = 19.00 \, \text{g/mol} \)
Molar mass of \( \text{FeF}_3 = 55.85 + 3\times19.00 = 55.85 + 57.00 = 112.85 \, \text{g/mol} \)
Step 2: Calculate moles of \( \text{FeF}_3 \)
Given mass of \( \text{FeF}_3 = 89 \, \text{g} \)
Moles of \( \text{FeF}_3 = \frac{\text{mass}}{\text{molar mass}} = \frac{89}{112.85} \approx 0.789 \, \text{mol} \)
Step 3: Determine moles of \( \text{F}_2 \) from stoichiometry
From the reaction, \( 2 \, \text{mol} \, \text{FeF}_3 \) is produced from \( 3 \, \text{mol} \, \text{F}_2 \)
So, moles of \( \text{F}_2 = \frac{3}{2} \times \text{moles of } \text{FeF}_3 = \frac{3}{2} \times 0.789 \approx 1.1835 \, \text{mol} \)
Step 4: Calculate mass of \( \text{F}_2 \)
Molar mass of \( \text{F}_2 = 2\times19.00 = 38.00 \, \text{g/mol} \)
Mass of \( \text{F}_2 = \text{moles} \times \text{molar mass} = 1.1835 \times 38.00 \approx 44.97 \, \text{g} \approx 45 \, \text{g} \) (rounded to a reasonable value)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Approximately 45 grams (or more precisely around 44.97 grams) of \( \text{F}_2 \) are needed.