QUESTION IMAGE
Question
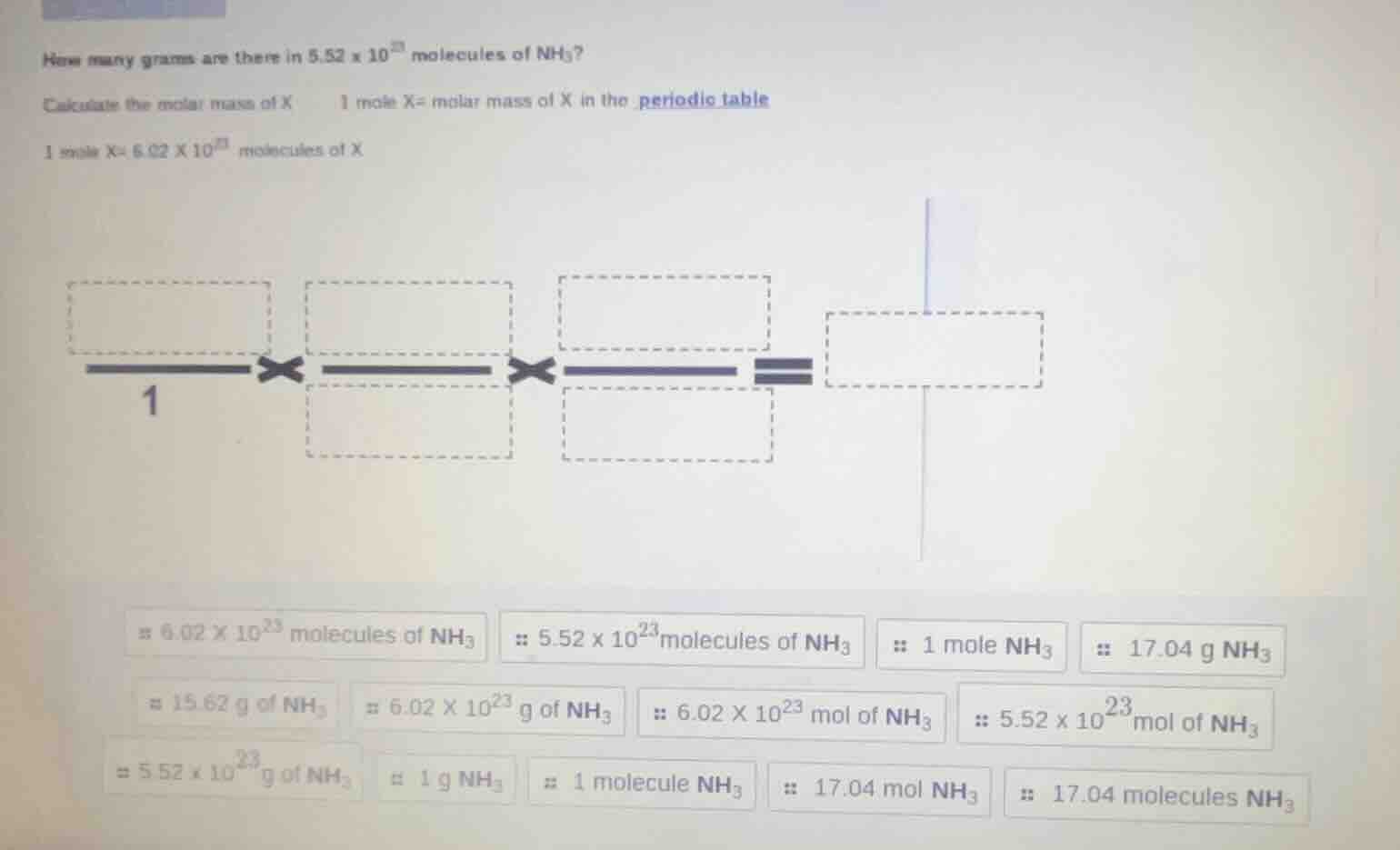
how many grams are there in $5.52 \times 10^{23}$ molecules of $\ce{nh_3}$?
calculate the molar mass of x \quad 1 mole x= molar mass of x in the periodic table
1 mole x= $6.02 \times 10^{23}$ molecules of x
image of a conversion factor setup with boxes to fill
$\boldsymbol{\\#\\ 6.02 \times 10^{23}}$ molecules of $\ce{nh_3}$ \quad $\boldsymbol{\because\\ 5.52 \times 10^{23}}$ molecules of $\ce{nh_3}$ \quad $\boldsymbol{\because\\ 1}$ mole $\ce{nh_3}$ \quad $\boldsymbol{\because\\ 17.04}$ g $\ce{nh_3}$
$\boldsymbol{\approx 15.62}$ g of $\ce{nh_3}$ \quad $\boldsymbol{\approx 6.02 \times 10^{23}}$ g of $\ce{nh_3}$ \quad $\boldsymbol{\because\\ 6.02 \times 10^{23}}$ mol of $\ce{nh_3}$ \quad $\boldsymbol{\because\\ 5.52 \times 10^{23}}$ mol of $\ce{nh_3}$
$\boldsymbol{\approx 5.52 \times 10^{23}}$ g of $\ce{nh_3}$ \quad $\boldsymbol{\approx 1}$ g $\ce{nh_3}$ \quad $\boldsymbol{\because\\ 1}$ molecule $\ce{nh_3}$ \quad $\boldsymbol{\because\\ 17.04}$ mol $\ce{nh_3}$ \quad $\boldsymbol{\because\\ 17.04}$ molecules $\ce{nh_3}$
Step 1: Convert molecules to moles
We know that \(1\) mole of \(NH_3\) contains \(6.02\times10^{23}\) molecules. So, to find the number of moles of \(NH_3\) in \(5.52\times 10^{23}\) molecules, we use the conversion factor \(\frac{1\space mole\space NH_3}{6.02\times 10^{23}\space molecules\space NH_3}\).
The number of moles \(n=\frac{5.52\times 10^{23}\space molecules\space NH_3}{6.02\times 10^{23}\space molecules\space NH_3/mol}\)
Step 2: Convert moles to grams
The molar mass of \(NH_3\) is \(17.04\space g/mol\) (since atomic mass of \(N = 14.01\space g/mol\) and \(H=1.01\space g/mol\), so \(14.01 + 3\times1.01=17.04\space g/mol\)). Now, we multiply the number of moles by the molar mass to get the mass in grams.
Mass \(m=n\times17.04\space g/mol=\frac{5.52\times 10^{23}}{6.02\times 10^{23}}\times17.04\)
First, calculate \(\frac{5.52}{6.02}\approx0.917\)
Then, \(0.917\times17.04\approx15.62\space g\)
Alternatively, using the factor - label method directly:
\(5.52\times 10^{23}\space molecules\space NH_3\times\frac{1\space mole\space NH_3}{6.02\times 10^{23}\space molecules\space NH_3}\times\frac{17.04\space g\space NH_3}{1\space mole\space NH_3}\)
\(=\frac{5.52\times 10^{23}\times17.04}{6.02\times 10^{23}}\space g\)
The \(10^{23}\) terms cancel out, and we have \(\frac{5.52\times17.04}{6.02}\space g\)
\(5.52\times17.04 = 5.52\times(17 + 0.04)=5.52\times17+5.52\times0.04 = 93.84+0.2208 = 94.0608\)
Then \(\frac{94.0608}{6.02}\approx15.62\space g\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The mass of \(5.52\times 10^{23}\) molecules of \(NH_3\) is approximately \(15.62\space g\)